A prospective study on drug monitoring of PEGasparaginase and Erwinia asparaginase and asparaginase antibodies in pediatric acute lymphoblastic leukemia
- PMID: 24449211
- PMCID: PMC3968389
- DOI: 10.1182/blood-2013-10-534347
A prospective study on drug monitoring of PEGasparaginase and Erwinia asparaginase and asparaginase antibodies in pediatric acute lymphoblastic leukemia
Abstract
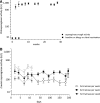
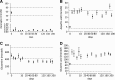
This study prospectively analyzed the efficacy of very prolonged courses of pegylated Escherichia coli asparaginase (PEGasparaginase) and Erwinia asparaginase in pediatric acute lymphoblastic leukemia (ALL) patients. Patients received 15 PEGasparaginase infusions (2500 IU/m(2) every 2 weeks) in intensification after receiving native E coli asparaginase in induction. In case of allergy to or silent inactivation of PEGasparaginase, Erwinia asparaginase (20 000 IU/m(2) 2-3 times weekly) was given. Eighty-nine patients were enrolled in the PEGasparaginase study. Twenty (22%) of the PEGasparaginase-treated patients developed an allergy; 7 (8%) showed silent inactivation. The PEGasparaginase level was 0 in all allergic patients (grade 1-4). Patients without hypersensitivity to PEGasparaginase had serum mean trough levels of 899 U/L. Fifty-nine patients were included in the Erwinia asparaginase study; 2 (3%) developed an allergy and none silent inactivation. Ninety-six percent had at least 1 trough level ≥100 U/L. The serum asparagine level was not always completely depleted with Erwinia asparaginase in contrast to PEGasparaginase. The presence of asparaginase antibodies was related to allergies and silent inactivation, but with low specificity (64%). Use of native E coli asparaginase in induction leads to high hypersensitivity rates to PEGasparaginase in intensification. Therefore, PEGasparaginase should be used upfront in induction, and we suggest that the dose could be lowered. Switching to Erwinia asparaginase leads to effective asparaginase levels in most patients. Therapeutic drug monitoring has been added to our ALL-11 protocol to individualize asparaginase therapy.
Figures



Comment in
-
Shedding light on the asparaginase galaxy.Blood. 2014 Mar 27;123(13):1976-8. doi: 10.1182/blood-2014-02-553040. Blood. 2014. PMID: 24677398
References
-
- Pui CH, Evans WE. Treatment of acute lymphoblastic leukemia. N Engl J Med. 2006;354(2):166–178. - PubMed
-
- Bussolati O, Belletti S, Uggeri J, et al. Characterization of apoptotic phenomena induced by treatment with L-asparaginase in NIH3T3 cells. Exp Cell Res. 1995;220(2):283–291. - PubMed
-
- Stams WA, den Boer ML, Beverloo HB, et al. Sensitivity to L-asparaginase is not associated with expression levels of asparagine synthetase in t(12;21)+ pediatric ALL. Blood. 2003;101(7):2743–2747. - PubMed
-
- Amylon MD, Shuster J, Pullen J, et al. Intensive high-dose asparaginase consolidation improves survival for pediatric patients with T cell acute lymphoblastic leukemia and advanced stage lymphoblastic lymphoma: a Pediatric Oncology Group study. Leukemia. 1999;13(3):335–342. - PubMed
-
- Silverman LB, Gelber RD, Dalton VK, et al. Improved outcome for children with acute lymphoblastic leukemia: results of Dana-Farber Consortium Protocol 91-01. Blood. 2001;97(5):1211–1218. - PubMed

