An attenuated duck plague virus (DPV) vaccine induces both systemic and mucosal immune responses to protect ducks against virulent DPV infection
- PMID: 24451329
- PMCID: PMC3993120
- DOI: 10.1128/CVI.00605-13
An attenuated duck plague virus (DPV) vaccine induces both systemic and mucosal immune responses to protect ducks against virulent DPV infection
Abstract
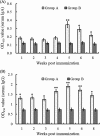
Duck plague (DP) is a severe disease caused by DP virus (DPV). Control of the disease is recognized as one of the biggest challenges in avian medicine. Vaccination is an efficient way to control DPV, and an attenuated vaccine is the main routine vaccine. The attenuated DPV vaccine strain CHa is a modified live vaccine, but the systemic and mucosal immune responses induced by this vaccine have been poorly understood. In this study, the immunogenicity and efficacy of the vaccine were evaluated after subcutaneous immunization of ducks. CD4(+) and CD8(+) T cells were counted by flow cytometry, and humoral and mucosal Ig antibodies were analyzed by enzyme-linked immunosorbent assay (ELISA). The results showed that high levels of T cells and Ig antibodies were present postimmunization and that there were more CD4(+) T cells than CD8(+) T cells. Titers of humoral IgG were higher than those of humoral IgA. Local IgA was found in each sample, whereas local IgG was found only in the spleen, thymus, bursa of Fabricius, harderian gland, liver, bile, and lung. In a protection assay, the attenuated DPV vaccine completely protected ducks against 1,000 50% lethal doses (LD50) of the lethal DPV strain CHv via oral infection. These data suggest that this subcutaneous vaccine elicits sufficient systemic and mucosal immune responses against lethal DPV challenge to be protective in ducks. This study provides broad insights into understanding the immune responses to the attenuated DPV vaccine strain CHa through subcutaneous immunization in ducks.
Figures



Similar articles
-
RNA-seq comparative analysis of Peking ducks spleen gene expression 24 h post-infected with duck plague virulent or attenuated virus.Vet Res. 2017 Sep 13;48(1):47. doi: 10.1186/s13567-017-0456-z. Vet Res. 2017. PMID: 28903751 Free PMC article.
-
Isolation and Selection of Duck Primary Cells as Pathogenic and Innate Immunologic Cell Models for Duck Plague Virus.Front Immunol. 2020 Jan 28;10:3131. doi: 10.3389/fimmu.2019.03131. eCollection 2019. Front Immunol. 2020. PMID: 32063900 Free PMC article.
-
Efficacy of Recombinant HVT-IBD Vaccines Administered to Broiler Chicks from a Single Breeder Flock at 30 and 60 Weeks of Age.Avian Dis. 2016 Sep;60(3):603-12. doi: 10.1637/11344-120815-Reg.1. Avian Dis. 2016. PMID: 27610719 Clinical Trial.
-
Sublingual immunization with Japanese encephalitis virus vaccine effectively induces immunity through both cellular and humoral immune responses in mice.Microbiol Immunol. 2016 Dec;60(12):846-853. doi: 10.1111/1348-0421.12458. Microbiol Immunol. 2016. PMID: 28004418
-
Comparison of the immune responses in BALB/c mice following immunization with DNA-based and live attenuated vaccines delivered via different routes.Vaccine. 2013 Feb 18;31(9):1353-6. doi: 10.1016/j.vaccine.2012.09.009. Epub 2012 Sep 17. Vaccine. 2013. PMID: 22995858
Cited by
-
A Recombinant Duck Plague Virus Containing the ICP27 Deletion Marker Provides Robust Protection in Ducks.Microbiol Spectr. 2023 Aug 17;11(4):e0098323. doi: 10.1128/spectrum.00983-23. Epub 2023 Jul 5. Microbiol Spectr. 2023. PMID: 37404171 Free PMC article.
-
Pathogenicity and transmissibility studies on live attenuated duck enteritis virus vaccine in non-target species.Front Microbiol. 2022 Nov 10;13:979368. doi: 10.3389/fmicb.2022.979368. eCollection 2022. Front Microbiol. 2022. PMID: 36439841 Free PMC article.
-
Molecular characterization of duck plague virus from selected Haor areas of Bangladesh.Open Vet J. 2021 Jan-Mar;11(1):42-51. doi: 10.4314/ovj.v11i1.8. Epub 2021 Jan 23. Open Vet J. 2021. PMID: 33898283 Free PMC article.
-
Duck plague virus UL47 gene affects the release and cell-to-cell spread of the virus and its deletion strains can provide strong protection for ducks.Poult Sci. 2025 Jun;104(6):105092. doi: 10.1016/j.psj.2025.105092. Epub 2025 Mar 24. Poult Sci. 2025. PMID: 40158279 Free PMC article.
-
An efficient bivalent vaccine against duck Tembusu and duck plague viruses based on ribosomal skipping of DHAV-1 2A1 and abundant expression of DPV protein.Poult Sci. 2025 Aug;104(8):105317. doi: 10.1016/j.psj.2025.105317. Epub 2025 May 17. Poult Sci. 2025. PMID: 40424885 Free PMC article.
References
-
- Baudet A. 1923. Mortality in ducks in the Netherlands caused by a filterable virus; fowl plague. Tijdschr. Diergeneeskd. 50:455–459
-
- Cheng AC, Wang MS, Liu F. 2004. The preliminary application of PCR in research of clinical diagnosis and mechanisms of immunity and pathology of duck plague virus. Chin. J. Virol. 20:364–370
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

