Targeting arginine-dependent cancers with arginine-degrading enzymes: opportunities and challenges
- PMID: 24453997
- PMCID: PMC3893322
- DOI: 10.4143/crt.2013.45.4.251
Targeting arginine-dependent cancers with arginine-degrading enzymes: opportunities and challenges
Abstract
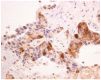
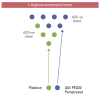
Arginine deprivation is a novel antimetabolite strategy for the treatment of arginine-dependent cancers that exploits differential expression and regulation of key urea cycle enzymes. Several studies have focused on inactivation of argininosuccinate synthetase 1 (ASS1) in a range of malignancies, including melanoma, hepatocellular carcinoma (HCC), mesothelial and urological cancers, sarcomas, and lymphomas. Epigenetic silencing has been identified as a key mechanism for loss of the tumor suppressor role of ASS1 leading to tumoral dependence on exogenous arginine. More recently, dysregulation of argininosuccinate lyase has been documented in a subset of arginine auxotrophic glioblastoma multiforme, HCC and in fumarate hydratase-mutant renal cancers. Clinical trials of several arginine depletors are ongoing, including pegylated arginine deiminase (ADI-PEG20, Polaris Group) and bioengineered forms of human arginase. ADI-PEG20 is furthest along the path of clinical development from combinatorial phase 1 to phase 3 trials and is described in more detail. The challenge will be to identify tumors sensitive to drugs such as ADI-PEG20 and integrate these agents into multimodality drug regimens using imaging and tissue/fluid-based biomarkers as predictors of response. Lastly, resistance pathways to arginine deprivation require further study to optimize arginine-targeted therapies in the oncology clinic.
Keywords: ADI-PEG20; Arginase; Arginine; Argininosuccinate lyase; Argininosuccinate synthetase; Drug combinations; Neoplasms.
Conflict of interest statement
Conflict of interest relevant to this article was not reported.
Figures
References
-
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. - PubMed
-
- Schiffman JD. No child left behind in SDHB testing for paragangliomas and pheochromocytomas. J Clin Oncol. 2011;29:4070–4072. - PubMed
-
- Vander Heiden MG. Targeting cancer metabolism: a therapeutic window opens. Nat Rev Drug Discov. 2011;10:671–684. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

