Regulation of skeletal muscle oxidative capacity and muscle mass by SIRT3
- PMID: 24454908
- PMCID: PMC3893254
- DOI: 10.1371/journal.pone.0085636
Regulation of skeletal muscle oxidative capacity and muscle mass by SIRT3
Abstract
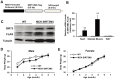
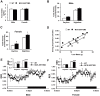
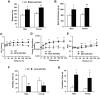
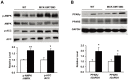
We have previously reported that the expression of mitochondrial deacetylase SIRT3 is high in the slow oxidative muscle and that the expression of muscle SIRT3 level is increased by dietary restriction or exercise training. To explore the function of SIRT3 in skeletal muscle, we report here the establishment of a transgenic mouse model with muscle-specific expression of the murine SIRT3 short isoform (SIRT3M3). Calorimetry study revealed that the transgenic mice had increased energy expenditure and lower respiratory exchange rate (RER), indicating a shift towards lipid oxidation for fuel usage, compared to control mice. The transgenic mice exhibited better exercise performance on treadmills, running 45% further than control animals. Moreover, the transgenic mice displayed higher proportion of slow oxidative muscle fibers, with increased muscle AMPK activation and PPARδ expression, both of which are known regulators promoting type I muscle fiber specification. Surprisingly, transgenic expression of SIRT3M3 reduced muscle mass up to 30%, likely through an up-regulation of FOXO1 transcription factor and its downstream atrophy gene MuRF-1. In summary, these results suggest that SIRT3 regulates the formation of oxidative muscle fiber, improves muscle metabolic function, and reduces muscle mass, changes that mimic the effects of caloric restriction.
Conflict of interest statement
Figures

Similar articles
-
Diet and exercise signals regulate SIRT3 and activate AMPK and PGC-1alpha in skeletal muscle.Aging (Albany NY). 2009 Aug 15;1(9):771-83. doi: 10.18632/aging.100075. Aging (Albany NY). 2009. PMID: 20157566 Free PMC article.
-
Loss of ovarian function in mice results in abrogated skeletal muscle PPARdelta and FoxO1-mediated gene expression.Biochem Biophys Res Commun. 2010 Jan 29;392(1):1-3. doi: 10.1016/j.bbrc.2009.10.072. Epub 2009 Oct 20. Biochem Biophys Res Commun. 2010. PMID: 19850007 Free PMC article.
-
Sirtuin-3 (Sirt3) regulates skeletal muscle metabolism and insulin signaling via altered mitochondrial oxidation and reactive oxygen species production.Proc Natl Acad Sci U S A. 2011 Aug 30;108(35):14608-13. doi: 10.1073/pnas.1111308108. Epub 2011 Aug 22. Proc Natl Acad Sci U S A. 2011. PMID: 21873205 Free PMC article.
-
Skeletal muscle overexpression of short isoform Sirt3 altered mitochondrial cardiolipin content and fatty acid composition.J Bioenerg Biomembr. 2018 Apr;50(2):131-142. doi: 10.1007/s10863-018-9752-1. Epub 2018 Mar 27. J Bioenerg Biomembr. 2018. PMID: 29589261
-
SIRT3 and mitochondrial metabolism in neurodegenerative diseases.Neurochem Int. 2017 Oct;109:184-192. doi: 10.1016/j.neuint.2017.04.012. Epub 2017 Apr 25. Neurochem Int. 2017. PMID: 28449871 Review.
Cited by
-
SIRT3 as a Regulator of Non-alcoholic Fatty Liver Disease.J Lifestyle Med. 2014 Sep;4(2):80-5. doi: 10.15280/jlm.2014.4.2.80. Epub 2014 Sep 30. J Lifestyle Med. 2014. PMID: 26064858 Free PMC article. Review.
-
Vitamin D3 Exerts Beneficial Effects on C2C12 Myotubes through Activation of the Vitamin D Receptor (VDR)/Sirtuins (SIRT)1/3 Axis.Nutrients. 2023 Nov 7;15(22):4714. doi: 10.3390/nu15224714. Nutrients. 2023. PMID: 38004107 Free PMC article.
-
Analysis of Promoter Methylation of the Bovine FOXO1 Gene and Its Effect on Proliferation and Differentiation of Myoblasts.Animals (Basel). 2023 Jan 16;13(2):319. doi: 10.3390/ani13020319. Animals (Basel). 2023. PMID: 36670858 Free PMC article.
-
AAV-mediated Sirt1 overexpression in skeletal muscle activates oxidative capacity but does not prevent insulin resistance.Mol Ther Methods Clin Dev. 2016 Nov 16;5:16072. doi: 10.1038/mtm.2016.72. eCollection 2016. Mol Ther Methods Clin Dev. 2016. PMID: 27909699 Free PMC article.
-
Exercise enhancement by RGS14 disruption is mediated by brown adipose tissue.Aging Cell. 2023 Apr;22(4):e13791. doi: 10.1111/acel.13791. Epub 2023 Mar 10. Aging Cell. 2023. PMID: 36905127 Free PMC article.
References
-
- Fernandes G, Yunis EJ, Good RA (1976) Suppression of adenocarcinoma by the immunological consequences of calorie restriction. Nature 263: 504–507. - PubMed
-
- Hansen BC, Bodkin NL (1993) Primary prevention of diabetes mellitus by prevention of obesity in monkeys. Diabetes 42: 1809–1814. - PubMed
-
- Hepple RT, Baker DJ, Kaczor JJ, Krause DJ (2005) Long-term caloric restriction abrogates the age-related decline in skeletal muscle aerobic function. FASEB J 19: 1320–1322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

