Characterization of angiotensin-converting enzyme 2 ectodomain shedding from mouse proximal tubular cells
- PMID: 24454948
- PMCID: PMC3893316
- DOI: 10.1371/journal.pone.0085958
Characterization of angiotensin-converting enzyme 2 ectodomain shedding from mouse proximal tubular cells
Abstract
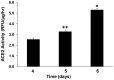
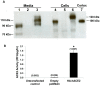
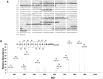
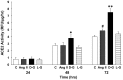
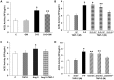
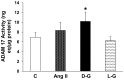
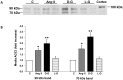
Angiotensin-converting enzyme 2 (ACE2) is highly expressed in the kidney proximal tubule, where it cleaves angiotensin (Ang) II to Ang-(1-7). Urinary ACE2 levels increase in diabetes, suggesting that ACE2 may be shed from tubular cells. The aim of this study was to determine if ACE2 is shed from proximal tubular cells, to characterize ACE2 fragments, and to study pathways for shedding. Studies involved primary cultures of mouse proximal tubular cells, with ACE2 activity measured using a synthetic substrate, and analysis of ACE2 fragments by immunoblots and mass spectrometry. The culture media from mouse proximal tubular cells demonstrated a time-dependent increase in ACE2 activity, suggesting constitutive ACE2 shedding. ACE2 was detected in media as two bands at ∼ 90 kDa and ∼ 70 kDa on immunoblots. By contrast, full-length ACE2 appeared at ∼ 100 kDa in cell lysates or mouse kidney cortex. Mass spectrometry of the two deglycosylated fragments identified peptides matching mouse ACE2 at positions 18-706 and 18-577, respectively. The C-terminus of the 18-706 peptide fragment contained a non-tryptic site, suggesting that Met(706) is a candidate ACE2 cleavage site. Incubation of cells in high D-glucose (25 mM) (and to a lesser extent Ang II) for 48-72 h increased ACE2 activity in the media (p<0.001), an effect blocked by inhibition of a disintegrin and metalloproteinase (ADAM)17. High D-glucose increased ADAM17 activity in cell lysates (p<0.05). These data indicate that two glycosylated ACE2 fragments are constitutively shed from mouse proximal tubular cells. ACE2 shedding is stimulated by high D-glucose, at least partly via an ADAM17-mediated pathway. The results suggest that proximal tubular shedding of ACE2 may increase in diabetes, which could enhance degradation of Ang II in the tubular lumen, and increase levels of Ang-(1-7).
Conflict of interest statement
Figures



Similar articles
-
Functional and molecular evidence for expression of the renin angiotensin system and ADAM17-mediated ACE2 shedding in COS7 cells.Am J Physiol Cell Physiol. 2015 May 1;308(9):C767-77. doi: 10.1152/ajpcell.00247.2014. Epub 2015 Mar 4. Am J Physiol Cell Physiol. 2015. PMID: 25740155 Free PMC article.
-
Insulin treatment attenuates renal ADAM17 and ACE2 shedding in diabetic Akita mice.Am J Physiol Renal Physiol. 2014 Mar 15;306(6):F629-39. doi: 10.1152/ajprenal.00516.2013. Epub 2014 Jan 22. Am J Physiol Renal Physiol. 2014. PMID: 24452639 Free PMC article.
-
Protein Kinase C-δ Mediates Shedding of Angiotensin-Converting Enzyme 2 from Proximal Tubular Cells.Front Pharmacol. 2016 Jun 1;7:146. doi: 10.3389/fphar.2016.00146. eCollection 2016. Front Pharmacol. 2016. PMID: 27313531 Free PMC article.
-
Angiotensin converting enzyme 2: a new important player in the regulation of glycemia.IUBMB Life. 2013 Sep;65(9):731-8. doi: 10.1002/iub.1190. Epub 2013 Jul 29. IUBMB Life. 2013. PMID: 23893738 Free PMC article. Review.
-
Angiotensin-converting enzyme 2: enhancing the degradation of angiotensin II as a potential therapy for diabetic nephropathy.Kidney Int. 2012 Mar;81(6):520-8. doi: 10.1038/ki.2011.381. Epub 2011 Nov 23. Kidney Int. 2012. PMID: 22113528 Review.
Cited by
-
COVID-19 Usurps Host Regulatory Networks.Front Pharmacol. 2020 Aug 14;11:1278. doi: 10.3389/fphar.2020.01278. eCollection 2020. Front Pharmacol. 2020. PMID: 32922297 Free PMC article. Review.
-
Response to recent commentaries regarding the involvement of angiotensin-converting enzyme 2 (ACE2) and renin-angiotensin system blockers in SARS-CoV-2 infections.Drug Dev Res. 2020 Sep;81(6):643-646. doi: 10.1002/ddr.21672. Epub 2020 Apr 17. Drug Dev Res. 2020. PMID: 32304146 Free PMC article. No abstract available.
-
ADAM10 and ADAM17, Major Regulators of Chronic Kidney Disease Induced Atherosclerosis?Int J Mol Sci. 2023 Apr 15;24(8):7309. doi: 10.3390/ijms24087309. Int J Mol Sci. 2023. PMID: 37108478 Free PMC article. Review.
-
Aortic Aneurysms, Chronic Kidney Disease and Metalloproteinases.Biomolecules. 2021 Jan 30;11(2):194. doi: 10.3390/biom11020194. Biomolecules. 2021. PMID: 33573220 Free PMC article. Review.
-
Glycemic control of type 2 diabetic patients with coronavirus disease during hospitalization: a proposal for early insulin therapy.Am J Physiol Endocrinol Metab. 2020 Jun 1;318(6):E835-E837. doi: 10.1152/ajpendo.00163.2020. Am J Physiol Endocrinol Metab. 2020. PMID: 32401039 Free PMC article. No abstract available.
References
-
- Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, et al. (2000) A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ Res 87: E1–9. - PubMed
-
- Tipnis SR, Hooper NM, Hyde R, Karran E, Christie G, et al. (2000) A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J Biol Chem 275: 33238–33243. - PubMed
-
- Ye M, Wysocki J, Naaz P, Salabat MR, LaPointe MS, et al. (2004) Increased ACE 2 and decreased ACE protein in renal tubules from diabetic mice: a renoprotective combination? Hypertension 43: 1120–1125. - PubMed
-
- Li N, Zimpelmann J, Cheng K, Wilkins JA, Burns KD (2005) The role of angiotensin converting enzyme 2 in the generation of angiotensin 1-7 by rat proximal tubules. Am J Physiol Renal Physiol 288: F353–362. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

