New insights into molecular mechanisms of diabetic kidney disease
- PMID: 24461730
- PMCID: PMC3932114
- DOI: 10.1053/j.ajkd.2013.10.047
New insights into molecular mechanisms of diabetic kidney disease
Abstract
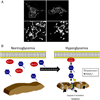
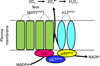
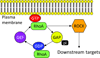
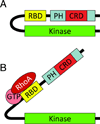
Diabetic kidney disease remains a major microvascular complication of diabetes and the most common cause of chronic kidney failure requiring dialysis in the United States. Medical advances over the past century have substantially improved the management of diabetes mellitus and thereby have increased patient survival. However, current standards of care reduce but do not eliminate the risk of diabetic kidney disease, and further studies are warranted to define new strategies for reducing the risk of diabetic kidney disease. In this review, we highlight some of the novel and established molecular mechanisms that contribute to the development of the disease and its outcomes. In particular, we discuss recent advances in our understanding of the molecular mechanisms implicated in the pathogenesis and progression of diabetic kidney disease, with special emphasis on the mitochondrial oxidative stress and microRNA targets. Additionally, candidate genes associated with susceptibility to diabetic kidney disease and alterations in various cytokines, chemokines, and growth factors are addressed briefly.
Keywords: End-stage renal disease (ESRD); diabetes mellitus; diabetic kidney disease; pathogenesis.
Copyright © 2014 National Kidney Foundation, Inc. Published by Elsevier Inc. All rights reserved.
Figures


References
-
- American Diabetes Association. Implications of the Diabetes Control and Complications Trial. Diabetes. 1993;42(11):1555–1558. - PubMed
-
- Remuzzi G, Ruggenenti P. Slowing the progression of diabetic kidney disease. N Engl J Med. 1993;329(20):1496–1497. - PubMed
-
- Lewis EJ, Hunsicker LG, Bain RP, Rohde RD. The effect of angiotensin-converting-enzyme inhibition on diabetic kidney disease. The Collaborative Study Group. N Engl J Med. 1993;329(20):1456–1462. - PubMed
-
- Writing Team for the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic kidney disease: the Epidemiology of Diabetes Interventions and Complications (EDIC) Study. JAMA. 2003;290(16):2159–2167. - PMC - PubMed
-
- Soldatos G, Cooper ME. Diabetic kidney disease: important pathophysiologic mechanisms. Diabetes Res Clin Pract. 2008;82(suppl 1):S75–S79. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

