The role of valvular endothelial cell paracrine signaling and matrix elasticity on valvular interstitial cell activation
- PMID: 24462357
- PMCID: PMC4040249
- DOI: 10.1016/j.biomaterials.2014.01.005
The role of valvular endothelial cell paracrine signaling and matrix elasticity on valvular interstitial cell activation
Abstract
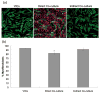
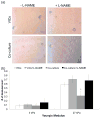
The effects of valvular endothelial cell (VlvEC) paracrine signaling on VIC phenotype and nodule formation were tested using a co-culture platform with physiologically relevant matrix elasticities and diffusion distance. 100 μm thin poly(ethylene glycol) (PEG) hydrogels of 3-27 kPa Young's moduli were fabricated in transwell inserts. VICs were cultured on the gels, as VIC phenotype is known to change significantly within this range, while VlvECs lined the underside of the membrane. Co-culture with VlvECs significantly reduced VIC activation to the myofibroblast phenotype on all gels with the largest percent decrease on the 3 kPa gels (~70%), while stiffer gels resulted in approximately 20-30% decrease. Additionally, VlvECs significantly reduced αSMA protein expression (~2 fold lower) on both 3 and 27 kPa gels, as well as the number (~2 fold lower) of nodules formed on the 27 kPa gels. Effects of VlvECs were prevented when nitric oxide (NO) release was inhibited with l-NAME, suggesting that VlvEC produced NO inhibits VIC activation. Withdrawal of l-NAME after 3, 5, and 7 days with restoration of VlvEC NO production for 2 additional days led to a partial reversal of VIC activation (~25% decrease). A potential mechanism by which VlvEC produced NO reduced VIC activation was studied by inhibiting initial and mid-stage cGMP pathway molecules. Inhibition of soluble guanylyl cyclase (sGC) with ODQ or protein kinase G (PKG) with RBrcGMP or stimulation of Rho kinase (ROCK) with LPA, abolished VlvEC effects on VIC activation. This work contributes substantially to the understanding of the valve endothelium's role in preventing VIC functions associated with aortic valve stenosis initiation and progression.
Keywords: Nitric oxide; PEG hydrogels; Valvular endothelial cells; Valvular interstitial cells; α-Smooth muscle actin.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures





References
-
- Freeman RV, Otto CM. Spectrum of calcific aortic valve disease pathogenesis, disease progression, and treatment strategies. Circulation. 2005;111:3316–3326. - PubMed
-
- Stewart BF, Siscovick D, Lind BK, Gardin JM, Gottdiener JS, Smith VE, et al. Clinical factors associated with calcific aortic valve disease. Cardiovascular Health Study. J Am Coll Cardiol. 1997;29:630–634. - PubMed
-
- Otto CM, Lind BK, Kitzman DW, Gersh BJ, Siscovick DS. Association of sortic-valve sclerosis with cardiovascular mortality and morbidity in the elderly. New Eng J Med. 1999;341:142–147. - PubMed
-
- Chester AH, Latif N, Yacoub MH, Taylor PM. Vascular complications in human disease, Chapter 18: Aortic Valve: From Function to Tissue Engineering. Springer; London: 2008.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

