Conditional inactivation of Miwi2 reveals that MIWI2 is only essential for prospermatogonial development in mice
- PMID: 24464225
- PMCID: PMC3978311
- DOI: 10.1038/cdd.2014.5
Conditional inactivation of Miwi2 reveals that MIWI2 is only essential for prospermatogonial development in mice
Abstract
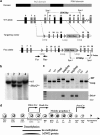
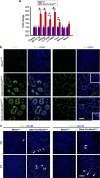
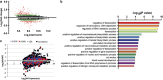
The PIWI-piRNA pathway serves as a critical defense mechanism through which the genome of the male germline is protected from invasion by transposable elements (TEs). MIWI2/PIWIL4, a member of the murine PIWI subclade of the Argonaute family, has been shown to be expressed in primordial germ cells (PGCs) and prospermatogonia in fetal and prepubertal testes. Global inactivation of Miwi2 leads to male sterility due to an early meiotic arrest, which correlates with retrotransposon desuppression. However, it remains unclear whether MIWI2 functions beyond the PGC stage and whether MIWI2 has a role beyond TE suppression during male germ line development. Through conditional inactivation of Miwi2, we demonstrate herein that MIWI2 function is restricted to a narrow time window during male PGC reprograming and that Miwi2 is dispensable for postnatal male germline development and testicular function in mice. Moreover, persistent activation of LINE1 and IAP retrotransposons caused by Miwi2 inactivation is compatible with mitotic cell cycle progression of spermatogonia during the first wave of spermatogenesis, but can cause zygotene to pachytene arrest in early meiosis due to multiple defects including enhanced DNA double-strand breaks, aberrant histone modifications and altered mRNA transcriptome. Our data not only validate those from global Miwi2 KO studies, but also suggest that MIWI2 and MIWI2-associated piRNAs have functions beyond TE suppression.
Figures




Similar articles
-
The endonuclease activity of Mili fuels piRNA amplification that silences LINE1 elements.Nature. 2011 Oct 23;480(7376):259-63. doi: 10.1038/nature10547. Nature. 2011. PMID: 22020280
-
MIWI2 and MILI Have Differential Effects on piRNA Biogenesis and DNA Methylation.Cell Rep. 2015 Aug 25;12(8):1234-43. doi: 10.1016/j.celrep.2015.07.036. Epub 2015 Aug 13. Cell Rep. 2015. PMID: 26279574 Free PMC article.
-
Miwi catalysis is required for piRNA amplification-independent LINE1 transposon silencing.Nature. 2011 Nov 27;480(7376):264-7. doi: 10.1038/nature10672. Nature. 2011. PMID: 22121019
-
piRNAs and PIWI proteins: regulators of gene expression in development and stem cells.Development. 2018 Sep 7;145(17):dev161786. doi: 10.1242/dev.161786. Development. 2018. PMID: 30194260 Review.
-
The regulatory functions of piRNA/PIWI in spermatogenesis.Yi Chuan. 2017 Aug 20;39(8):683-691. doi: 10.16288/j.yczz.17-245. Yi Chuan. 2017. PMID: 28903896 Review.
Cited by
-
Genetic etiological spectrum of sperm morphological abnormalities.J Assist Reprod Genet. 2024 Nov;41(11):2877-2929. doi: 10.1007/s10815-024-03274-8. Epub 2024 Oct 17. J Assist Reprod Genet. 2024. PMID: 39417902 Review.
-
Themes and variations on piRNA-guided transposon control.Mob DNA. 2023 Sep 2;14(1):10. doi: 10.1186/s13100-023-00298-2. Mob DNA. 2023. PMID: 37660099 Free PMC article. Review.
-
UPF2, a nonsense-mediated mRNA decay factor, is required for prepubertal Sertoli cell development and male fertility by ensuring fidelity of the transcriptome.Development. 2015 Jan 15;142(2):352-62. doi: 10.1242/dev.115642. Epub 2014 Dec 11. Development. 2015. PMID: 25503407 Free PMC article.
-
Somatic Miwi2 modulates mitochondrial function in airway multiciliated cells and exacerbates influenza pathogenesis.iScience. 2025 Mar 25;28(4):112291. doi: 10.1016/j.isci.2025.112291. eCollection 2025 Apr 18. iScience. 2025. PMID: 40241756 Free PMC article.
-
UPF2-Dependent Nonsense-Mediated mRNA Decay Pathway Is Essential for Spermatogenesis by Selectively Eliminating Longer 3'UTR Transcripts.PLoS Genet. 2016 May 5;12(5):e1005863. doi: 10.1371/journal.pgen.1005863. eCollection 2016 May. PLoS Genet. 2016. PMID: 27149259 Free PMC article.
References
-
- Hutvagner G, Simard MJ. Argonaute proteins: key players in RNA silencing. Nat Rev. Mol Cell Biol. 2008;9:22–32. - PubMed
-
- Girard A, Sachidanandam R, Hannon GJ, Carmell MA. A germline-specific class of small RNAs binds mammalian Piwi proteins. Nature. 2006;442:199–202. - PubMed
-
- Lau NC, Seto AG, Kim J, Kuramochi-Miyagawa S, Nakano T, Bartel DP, et al. Characterization of the piRNA complex from rat testes. Science. 2006;313:363–367. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

