Comparative analysis of αB-crystallin expression in heat-stressed myocardial cells in vivo and in vitro
- PMID: 24466295
- PMCID: PMC3899361
- DOI: 10.1371/journal.pone.0086937
Comparative analysis of αB-crystallin expression in heat-stressed myocardial cells in vivo and in vitro
Abstract
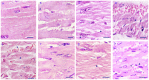
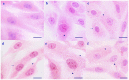
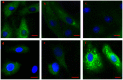
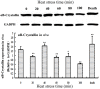
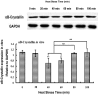
Relationships between αB-crystallin expression patterns and pathological changes of myocardial cells after heat stress were examined in vitro and in vivo in this study using the H9C2 cell line and Sprague-Dawley rats, respectively. Histopathological lesions, characterized by acute degeneration, karyopyknosis and loss of a defined nucleus, became more severe in rat hearts over the course of heat stress treatment from 20 min to 100 min. The expression of αB-crystallin in rat hearts showed a significant decrease (P<0.05) throughout the heat stress treatment period, except at the 40 min time point. Likewise, decreased αB-crystallin expression was also observed in the H9C2 cell line exposed to a high temperature in vitro, although its expression recovered to normal levels at later time points (80 and 100 min) and the cellular damage was less severe. The results suggest that αB-crystallin is mobilized early after exposure to a high temperature to interact with damaged proteins but that the myocardial cells cannot produce sufficient αB-crystallin for protection against heat stress. Lower αB-crystallin expression levels were accompanied by obvious cell/tissue damage, suggesting that the abundance of this protein is associated with protective effects in myocardial cells in vitro and in vivo. Thus, αB-crystallin is a potential biomarker of heat stress.
Conflict of interest statement
Figures

Similar articles
-
Expression profiles of heat shock protein 27 and αB-crystallin and their effects on heat-stressed rat myocardial cells in vitro and in vivo.Mol Med Rep. 2016 Feb;13(2):1633-8. doi: 10.3892/mmr.2015.4693. Epub 2015 Dec 17. Mol Med Rep. 2016. PMID: 26708692
-
Localization and expression of Hsp27 and αB-crystallin in rat primary myocardial cells during heat stress in vitro.PLoS One. 2013 Jul 19;8(7):e69066. doi: 10.1371/journal.pone.0069066. Print 2013. PLoS One. 2013. PMID: 23894407 Free PMC article.
-
O-GlcNAcylation of αB-crystallin regulates its stress-induced translocation and cytoprotection.Mol Cell Biochem. 2013 Jul;379(1-2):59-68. doi: 10.1007/s11010-013-1627-5. Epub 2013 Mar 30. Mol Cell Biochem. 2013. PMID: 23543138
-
αB-crystallin as a promising target in pathological conditions - A review.Ann Agric Environ Med. 2020 Sep 11;27(3):326-334. doi: 10.26444/aaem/111759. Epub 2019 Sep 6. Ann Agric Environ Med. 2020. PMID: 32955210 Review.
-
Alpha crystallins in the retinal pigment epithelium and implications for the pathogenesis and treatment of age-related macular degeneration.Biochim Biophys Acta. 2016 Jan;1860(1 Pt B):258-68. doi: 10.1016/j.bbagen.2015.05.016. Epub 2015 May 27. Biochim Biophys Acta. 2016. PMID: 26026469 Free PMC article. Review.
Cited by
-
Differential Glycosylation and Modulation of Camel and Human HSP Isoforms in Response to Thermal and Hypoxic Stresses.Int J Mol Sci. 2018 Jan 30;19(2):402. doi: 10.3390/ijms19020402. Int J Mol Sci. 2018. PMID: 29385708 Free PMC article.
-
Modulation of Heat-Shock Proteins Mediates Chicken Cell Survival against Thermal Stress.Animals (Basel). 2020 Dec 16;10(12):2407. doi: 10.3390/ani10122407. Animals (Basel). 2020. PMID: 33339245 Free PMC article. Review.
-
Cytoprotective effect of palm kernel cake phenolics against aflatoxin B1-induced cell damage and its underlying mechanism of action.BMC Complement Altern Med. 2015 Oct 30;15:392. doi: 10.1186/s12906-015-0921-z. BMC Complement Altern Med. 2015. PMID: 26518905 Free PMC article.
-
Chronic heat stress induces the expression of HSP genes in the retina of chickens (Gallus gallus).Front Genet. 2023 Apr 3;14:1085590. doi: 10.3389/fgene.2023.1085590. eCollection 2023. Front Genet. 2023. PMID: 37077545 Free PMC article.
-
A monoclonal antibody targeted to the functional peptide of αB-crystallin inhibits the chaperone and anti-apoptotic activities.J Immunol Methods. 2019 Apr;467:37-47. doi: 10.1016/j.jim.2019.02.004. Epub 2019 Feb 6. J Immunol Methods. 2019. PMID: 30738041 Free PMC article.
References
-
- Bettaieb A, Averill-Bates DA (2008) Thermotolerance induced at a fever temperature of 40 degrees C protects cells against hyperthermia-induced apoptosis mediated by death receptor signalling. Biochem Cell Biol 86: 521–538. - PubMed
-
- Rosenthal A, Button LN, Nathan DG, Miettinen OS, Nadas AS (1971) Blood volume changes in cyanotic congenital heart disease. Am J Cardiol 27: 162–167. - PubMed
-
- Bouchama A, Hammami MM, Haq A, Jackson J, al-Sedairy S (1996) Evidence for endothelial cell activation/injury in heatstroke. Crit Care Med 24: 1173–1178. - PubMed
-
- Ellis FP (1972) Mortality from heat illness and heat-aggravated illness in the United States. Environ Res 5: 1–58. - PubMed
-
- Scheers-Masters JR, Schootman M, Thach BT (2004) Heat stress and sudden infant death syndrome incidence: a United States population epidemiologic study. Pediatrics 113: e586–592. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

