Progesterone receptor-B enhances estrogen responsiveness of breast cancer cells via scaffolding PELP1- and estrogen receptor-containing transcription complexes
- PMID: 24469035
- PMCID: PMC4112172
- DOI: 10.1038/onc.2013.579
Progesterone receptor-B enhances estrogen responsiveness of breast cancer cells via scaffolding PELP1- and estrogen receptor-containing transcription complexes
Abstract
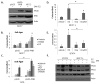
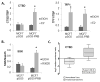
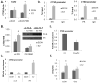
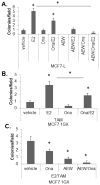
Progesterone and estrogen are important drivers of breast cancer proliferation. Herein, we probed estrogen receptor-α (ER) and progesterone receptor (PR) cross-talk in breast cancer models. Stable expression of PR-B in PR-low/ER+ MCF7 cells increased cellular sensitivity to estradiol and insulin-like growth factor 1 (IGF1), as measured in growth assays performed in the absence of exogenous progestin; similar results were obtained in PR-null/ER+ T47D cells stably expressing PR-B. Genome-wide microarray analyses revealed that unliganded PR-B induced robust expression of a subset of estradiol-responsive ER target genes, including cathepsin-D (CTSD). Estradiol-treated MCF7 cells stably expressing PR-B exhibited enhanced ER Ser167 phosphorylation and recruitment of ER, PR and the proline-, glutamate- and leucine-rich protein 1 (PELP1) to an estrogen response element in the CTSD distal promoter; this complex co-immunoprecipitated with IGF1 receptor (IGFR1) in whole-cell lysates. Importantly, ER/PR/PELP1 complexes were also detected in human breast cancer samples. Inhibition of IGF1R or phosphoinositide 3-kinase blocked PR-B-dependent CTSD mRNA upregulation in response to estradiol. Similarly, inhibition of IGF1R or PR significantly reduced ER recruitment to the CTSD promoter. Stable knockdown of endogenous PR or onapristone treatment of multiple unmodified breast cancer cell lines blocked estradiol-mediated CTSD induction, inhibited growth in soft agar and partially restored tamoxifen sensitivity of resistant cells. Further, combination treatment of breast cancer cells with both onapristone and IGF1R tyrosine kinase inhibitor AEW541 was more effective than either agent alone. In summary, unliganded PR-B enhanced proliferative responses to estradiol and IGF1 via scaffolding of ER-α/PELP1/IGF1R-containing complexes. Our data provide a strong rationale for targeting PR in combination with ER and IGF1R in patients with luminal breast cancer.
Figures



References
-
- Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005 May 14–20;365(9472):1687–717. - PubMed
-
- Musgrove EA, Sutherland RL. Biological determinants of endocrine resistance in breast cancer. Nat Rev Cancer. 2009 Sep;9(9):631–43. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

