Noninvasive positron emission tomography and fluorescence imaging of CD133+ tumor stem cells
- PMID: 24469819
- PMCID: PMC3926065
- DOI: 10.1073/pnas.1314189111
Noninvasive positron emission tomography and fluorescence imaging of CD133+ tumor stem cells
Abstract
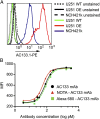
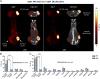
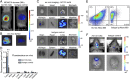
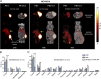
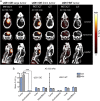
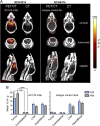
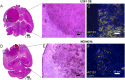
A technology that visualizes tumor stem cells with clinically relevant tracers could have a broad impact on cancer diagnosis and treatment. The AC133 epitope of CD133 currently is one of the best-characterized tumor stem cell markers for many intra- and extracranial tumor entities. Here we demonstrate the successful noninvasive detection of AC133(+) tumor stem cells by PET and near-infrared fluorescence molecular tomography in subcutaneous and orthotopic glioma xenografts using antibody-based tracers. Particularly, microPET with (64)Cu-NOTA-AC133 mAb yielded high-quality images with outstanding tumor-to-background contrast, clearly delineating subcutaneous tumor stem cell-derived xenografts from surrounding tissues. Intracerebral tumors as small as 2-3 mm also were clearly discernible, and the microPET images reflected the invasive growth pattern of orthotopic cancer stem cell-derived tumors with low density of AC133(+) cells. These data provide a basis for further preclinical and clinical use of the developed tracers for high-sensitivity and high-resolution monitoring of AC133(+) tumor stem cells.
Keywords: CSCs; cancer stem cells; glioblastoma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

