To gibberellins and beyond! Surveying the evolution of (di)terpenoid metabolism
- PMID: 24471837
- PMCID: PMC4118669
- DOI: 10.1146/annurev-arplant-050213-035705
To gibberellins and beyond! Surveying the evolution of (di)terpenoid metabolism
Abstract
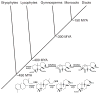
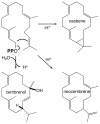
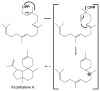
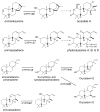
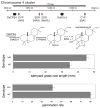
The diterpenoids are classically defined by their composition--four isoprenyl units (20 carbons)--and are generally derived from [E,E,E]-geranylgeranyl diphosphate (GGPP). Such metabolism seems to be ancient and has been extensively diversified, with ∼12,000 diterpenoid natural products known. Particularly notable are the gibberellin phytohormones, whose requisite biosynthesis has provided a genetic reservoir that gave rise to not only a large superfamily of ∼7,000 diterpenoids but also, to some degree, all plant terpenoid natural products. This review focuses on the diterpenoids, particularly the defining biosynthetic characteristics of the major superfamilies defined by the cyclization and/or rearrangement of GGPP catalyzed by diterpene synthases/cyclases, although it also includes some discussion of the important subsequent elaboration in the few cases where sufficient molecular genetic information is available. It additionally addresses the array of biological activity providing the selective pressures that drive the observed gene family expansion and diversification, along with biosynthetic gene clustering.
Figures

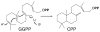
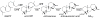



Similar articles
-
Divergent Evolution of the Diterpene Biosynthesis Pathway in Tea Plants (Camellia sinensis) Caused by Single Amino Acid Variation of ent-Kaurene Synthase.J Agric Food Chem. 2020 Sep 16;68(37):9930-9939. doi: 10.1021/acs.jafc.0c03488. Epub 2020 Sep 3. J Agric Food Chem. 2020. PMID: 32841021
-
Synergistic substrate inhibition of ent-copalyl diphosphate synthase: a potential feed-forward inhibition mechanism limiting gibberellin metabolism.Plant Physiol. 2007 May;144(1):445-54. doi: 10.1104/pp.106.095208. Epub 2007 Mar 23. Plant Physiol. 2007. PMID: 17384166 Free PMC article.
-
Diterpene synthases facilitating production of the kaurane skeleton of eriocalyxin B in the medicinal plant Isodon eriocalyx.Phytochemistry. 2019 Feb;158:96-102. doi: 10.1016/j.phytochem.2018.11.015. Epub 2018 Nov 26. Phytochemistry. 2019. PMID: 30496917
-
Evolution of Labdane-Related Diterpene Synthases in Cereals.Plant Cell Physiol. 2020 Dec 23;61(11):1850-1859. doi: 10.1093/pcp/pcaa106. Plant Cell Physiol. 2020. PMID: 32810270 Review.
-
Plant (di)terpenoid evolution: from pigments to hormones and beyond.Nat Prod Rep. 2023 Feb 22;40(2):452-469. doi: 10.1039/d2np00054g. Nat Prod Rep. 2023. PMID: 36472136 Free PMC article. Review.
Cited by
-
NDH expression marks major transitions in plant evolution and reveals coordinate intracellular gene loss.BMC Plant Biol. 2015 Apr 11;15:100. doi: 10.1186/s12870-015-0484-7. BMC Plant Biol. 2015. PMID: 25886915 Free PMC article.
-
Deceptive Complexity in Formation of Cleistantha-8,12-diene.Org Lett. 2022 Apr 15;24(14):2646-2649. doi: 10.1021/acs.orglett.2c00680. Epub 2022 Apr 6. Org Lett. 2022. PMID: 35385666 Free PMC article.
-
Direct production of dihydroxylated sesquiterpenoids by a maize terpene synthase.Plant J. 2018 Jun;94(5):847-856. doi: 10.1111/tpj.13901. Epub 2018 Apr 17. Plant J. 2018. PMID: 29570233 Free PMC article.
-
Gene refashioning through innovative shifting of reading frames in mosses.Nat Commun. 2018 Apr 19;9(1):1555. doi: 10.1038/s41467-018-04025-x. Nat Commun. 2018. PMID: 29674719 Free PMC article.
-
Enhancing plant growth and biomass production by overexpression of GA20ox gene under control of a root preferential promoter.Transgenic Res. 2022 Feb;31(1):73-85. doi: 10.1007/s11248-021-00282-7. Epub 2021 Sep 13. Transgenic Res. 2022. PMID: 34532833
References
-
- Akhtar TA, Matsuba Y, Schauvinhold I, Yu G, Lees HA, et al. The tomato cis-prenyltransferase gene family. Plant J. 2013;73:640–52. - PubMed
-
- Anterola A, Shanle E, Mansouri K, Schuette S, Renzaglia K. Gibberellin precursor is involved in spore germination in the moss Physcomitrella patens. Planta. 2009;229:1003–7. - PubMed
-
- Bomke C, Tudzynski B. Diversity, regulation, and evolution of the gibberellin biosynthetic pathway in fungi compared to plants and bacteria. Phytochemistry. 2009;70:1876–93. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

