TG-interacting factor 1 acts as a transcriptional repressor of sterol O-acyltransferase 2
- PMID: 24478032
- PMCID: PMC3966704
- DOI: 10.1194/jlr.M045922
TG-interacting factor 1 acts as a transcriptional repressor of sterol O-acyltransferase 2
Abstract
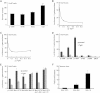
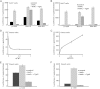
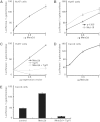
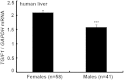
Acat2 [gene name: sterol O-acyltransferase 2 (SOAT2)] esterifies cholesterol in enterocytes and hepatocytes. This study aims to identify repressor elements in the human SOAT2 promoter and evaluate their in vivo relevance. We identified TG-interacting factor 1 (Tgif1) to function as an important repressor of SOAT2. Tgif1 could also block the induction of the SOAT2 promoter activity by hepatocyte nuclear factor 1α and 4α. Women have ∼ 30% higher hepatic TGIF1 mRNA compared with men. Depletion of Tgif1 in mice increased the hepatic Soat2 expression and resulted in higher hepatic lipid accumulation and plasma cholesterol levels. Tgif1 is a new player in human cholesterol metabolism.
Keywords: hepatocyte nuclear factor 1α and 4α; human; liver; triglyceride.
Figures
Similar articles
-
Overexpression of transforming growth factor β induced factor homeobox 1 represses NPC1L1 and lowers markers of intestinal cholesterol absorption.Atherosclerosis. 2018 Aug;275:246-255. doi: 10.1016/j.atherosclerosis.2018.06.867. Epub 2018 Jun 19. Atherosclerosis. 2018. PMID: 29980051
-
Role of TG-interacting factor (Tgif) in lipid metabolism.Biochim Biophys Acta. 2015 Jan;1851(1):9-12. doi: 10.1016/j.bbalip.2014.07.019. Epub 2014 Aug 1. Biochim Biophys Acta. 2015. PMID: 25088698 Review.
-
Control of ACAT2 liver expression by HNF1.J Lipid Res. 2005 Sep;46(9):1868-76. doi: 10.1194/jlr.M400450-JLR200. Epub 2005 Jun 16. J Lipid Res. 2005. PMID: 15961790
-
Soat2 ties cholesterol metabolism to β-oxidation and glucose tolerance in male mice.J Intern Med. 2022 Aug;292(2):296-307. doi: 10.1111/joim.13450. Epub 2022 Jan 13. J Intern Med. 2022. PMID: 34982494
-
Two novel cis-elements involved in hepatocyte nuclear factor 4α regulation of acyl-coenzyme A:cholesterol acyltransferase 2 expression.Acta Biochim Biophys Sin (Shanghai). 2012 Feb;44(2):162-71. doi: 10.1093/abbs/gmr102. Epub 2011 Dec 7. Acta Biochim Biophys Sin (Shanghai). 2012. PMID: 22155889
Cited by
-
Tgif1 and Tgif2 Repress Expression of the RabGAP Evi5l.Mol Cell Biol. 2017 Feb 15;37(5):e00527-16. doi: 10.1128/MCB.00527-16. Print 2017 Mar 1. Mol Cell Biol. 2017. PMID: 27956704 Free PMC article.
-
Structural Insight into the Binding of TGIF1 to SIN3A PAH2 Domain through a C-Terminal Amphipathic Helix.Int J Mol Sci. 2021 Nov 23;22(23):12631. doi: 10.3390/ijms222312631. Int J Mol Sci. 2021. PMID: 34884456 Free PMC article.
-
Establishment and characterization of cytochrome P450 1A1 CRISPR/Cas9 Knockout Bovine Foetal Hepatocyte Cell Line (BFH12).Cell Biol Toxicol. 2024 Mar 26;40(1):18. doi: 10.1007/s10565-024-09856-7. Cell Biol Toxicol. 2024. PMID: 38528259 Free PMC article.
-
Tgif1 and Tgif2 Regulate Axial Patterning in Mouse.PLoS One. 2016 May 17;11(5):e0155837. doi: 10.1371/journal.pone.0155837. eCollection 2016. PLoS One. 2016. PMID: 27187787 Free PMC article.
-
Transcriptome and co-expression network analysis reveals the molecular mechanism of inosine monophosphate-specific deposition in chicken muscle.Front Physiol. 2023 May 17;14:1199311. doi: 10.3389/fphys.2023.1199311. eCollection 2023. Front Physiol. 2023. PMID: 37265843 Free PMC article.
References
-
- Buhman K. K., Accad M., Novak S., Choi R. S., Wong J. S., Hamilton R. L., Turley S., Farese R. V., Jr 2000. Resistance to diet-induced hypercholesterolemia and gallstone formation in ACAT2-deficient mice. Nat. Med. 6: 1341–1347. - PubMed
-
- Smith J. L., Rangaraj K., Simpson R., Maclean D. J., Nathanson L. K., Stuart K. A., Scott S. P., Ramm G. A., de Jersey J. 2004. Quantitative analysis of the expression of ACAT genes in human tissues by real-time PCR. J. Lipid Res. 45: 686–696. - PubMed
-
- Parini P., Davis M., Lada A. T., Erickson S. K., Wright T. L., Gustafsson U., Sahlin S., Einarsson C., Eriksson M., Angelin B., et al. 2004. ACAT2 is localized to hepatocytes and is the major cholesterol-esterifying enzyme in human liver. Circulation. 110: 2017–2023. - PubMed
-
- Brown J. M., Bell T. A., III, Alger H. M., Sawyer J. K., Smith T. L., Kelley K., Shah R., Wilson M. D., Davis M. A., Lee R. G., et al. 2008. Targeted depletion of hepatic ACAT2-driven cholesterol esterification reveals a non-biliary route for fecal neutral sterol loss. J. Biol. Chem. 283: 10522–10534. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

