The dendritic location of the L-type current and its deactivation by the somatic AHP current both contribute to firing bistability in motoneurons
- PMID: 24478687
- PMCID: PMC3902208
- DOI: 10.3389/fncom.2014.00004
The dendritic location of the L-type current and its deactivation by the somatic AHP current both contribute to firing bistability in motoneurons
Abstract
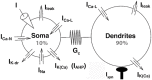
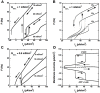
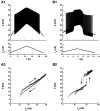
Spinal motoneurons may display a variety of firing patterns including bistability between repetitive firing and quiescence and, more rarely, bistability between two firing states of different frequencies. It was suggested in the past that firing bistability required that the persistent L-type calcium current be segregated in distal dendrites, far away from the spike generating currents. However, this is not supported by more recent data. Using a two compartment model of motoneuron, we show that the different firing patterns may also result from the competition between the more proximal dendritic component of the dendritic L-type conductance and the calcium sensitive potassium conductance responsible for afterhypolarization (AHP). Further emphasizing this point, firing bistability may be also achieved when the L-type current is put in the somatic compartment. However, this requires that the calcium-sensitive potassium conductance be triggered solely by the high threshold calcium currents activated during spikes and not by calcium influx through the L-type current. This prediction was validated by dynamic clamp experiments in vivo in lumbar motoneurons of deeply anesthetized cats in which an artificial L-type current was added at the soma. Altogether, our results suggest that the dynamical interaction between the L-type and afterhyperpolarization currents is as fundamental as the segregation of the calcium L-type current in dendrites for controlling the discharge of motoneurons.
Keywords: afterhyperpolarization; bistability; dynamic clamp; modeling; persistent calcium current.
Figures



Similar articles
-
Compartmental model of vertebrate motoneurons for Ca2+-dependent spiking and plateau potentials under pharmacological treatment.J Neurophysiol. 1997 Dec;78(6):3371-85. doi: 10.1152/jn.1997.78.6.3371. J Neurophysiol. 1997. PMID: 9405551
-
Dendritic L-type calcium currents in mouse spinal motoneurons: implications for bistability.Eur J Neurosci. 2000 May;12(5):1635-46. doi: 10.1046/j.1460-9568.2000.00055.x. Eur J Neurosci. 2000. PMID: 10792441
-
Accumulation of cytoplasmic calcium, but not apamin-sensitive afterhyperpolarization current, during high frequency firing in rat subthalamic nucleus cells.J Physiol. 2008 Feb 1;586(3):817-33. doi: 10.1113/jphysiol.2007.141929. Epub 2007 Dec 6. J Physiol. 2008. PMID: 18063664 Free PMC article.
-
Dendritic distributions of L-type Ca2+ and SKL channels in spinal motoneurons: a simulation study.J Neurophysiol. 2020 Oct 1;124(4):1285-1307. doi: 10.1152/jn.00169.2020. Epub 2020 Sep 16. J Neurophysiol. 2020. PMID: 32937080 Free PMC article.
-
Input-output functions of mammalian motoneurons.Rev Physiol Biochem Pharmacol. 2001;143:137-263. doi: 10.1007/BFb0115594. Rev Physiol Biochem Pharmacol. 2001. PMID: 11428264 Review.
Cited by
-
Tonic Activation of Extrasynaptic NMDA Receptors Decreases Intrinsic Excitability and Promotes Bistability in a Model of Neuronal Activity.Int J Mol Sci. 2019 Dec 27;21(1):206. doi: 10.3390/ijms21010206. Int J Mol Sci. 2019. PMID: 31892239 Free PMC article.
-
Frequency-Dependent Action of Neuromodulation.eNeuro. 2021 Nov 9;8(6):ENEURO.0338-21.2021. doi: 10.1523/ENEURO.0338-21.2021. Print 2021 Nov-Dec. eNeuro. 2021. PMID: 34593519 Free PMC article.
-
Contribution of intrinsic motoneuron properties to discharge hysteresis and its estimation based on paired motor unit recordings: a simulation study.J Neurophysiol. 2015 Jul;114(1):184-98. doi: 10.1152/jn.00019.2015. Epub 2015 Apr 22. J Neurophysiol. 2015. PMID: 25904704 Free PMC article.
-
Curated Model Development Using NEUROiD: A Web-Based NEUROmotor Integration and Design Platform.Front Neuroinform. 2019 Aug 7;13:56. doi: 10.3389/fninf.2019.00056. eCollection 2019. Front Neuroinform. 2019. PMID: 31440153 Free PMC article.
-
Swimming against the tide: investigations of the C-bouton synapse.Front Neural Circuits. 2014 Sep 18;8:106. doi: 10.3389/fncir.2014.00106. eCollection 2014. Front Neural Circuits. 2014. PMID: 25278842 Free PMC article. Review.
References
-
- Booth V., Rinzel J., Kiehn O. (1997). Compartmental model of vertebrate motoneurons for Ca2+-dependent spiking and plateau potentials under pharmacological treatment. J. Neurophysiol. 78, 3371–3385 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

