The recovery of acetylcholinesterase activity and the progression of neuropathological and pathophysiological alterations in the rat basolateral amygdala after soman-induced status epilepticus: relation to anxiety-like behavior
- PMID: 24486384
- PMCID: PMC4005290
- DOI: 10.1016/j.neuropharm.2014.01.035
The recovery of acetylcholinesterase activity and the progression of neuropathological and pathophysiological alterations in the rat basolateral amygdala after soman-induced status epilepticus: relation to anxiety-like behavior
Abstract
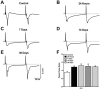
Organophosphorus nerve agents are powerful neurotoxins that irreversibly inhibit acetylcholinesterase (AChE) activity. One of the consequences of AChE inhibition is the generation of seizures and status epilepticus (SE), which cause brain damage, resulting in long-term neurological and behavioral deficits. Increased anxiety is the most common behavioral abnormality after nerve agent exposure. This is not surprising considering that the amygdala, and the basolateral nucleus of the amygdala (BLA) in particular, plays a central role in anxiety, and this structure suffers severe damage by nerve agent-induced seizures. In the present study, we exposed male rats to the nerve agent soman, at a dose that induce SE, and determined the time course of recovery of AChE activity, along with the progression of neuropathological and pathophysiological alterations in the BLA, during a 30-day period after exposure. Measurements were taken at 24 h, 7 days, 14 days, and 30 days after exposure, and at 14 and 30 days, anxiety-like behavior was also evaluated. We found that more than 90% of AChE is inhibited at the onset of SE, and AChE inhibition remains at this level 24 h later, in the BLA, as well as in the hippocampus, piriform cortex, and prelimbic cortex, which we analyzed for comparison. AChE activity recovered by day 7 in the BLA and day 14 in the other three regions. Significant neuronal loss and neurodegeneration were present in the BLA at 24 h and throughout the 30-day period. There was no significant loss of GABAergic interneurons in the BLA at 24 h post-exposure. However, by day 7, the number of GABAergic interneurons in the BLA was reduced, and at 14 and 30 days after soman, the ratio of GABAergic interneurons to the total number of neurons was lower compared to controls. Anxiety-like behavior in the open-field and the acoustic startle response tests was increased at 14 and 30 days post-exposure. Accompanying pathophysiological alterations in the BLA - studied in in vitro brain slices - included a reduction in the amplitude of field potentials evoked by stimulation of the external capsule, along with prolongation of their time course and an increase in the paired-pulse ratio. Long-term potentiation was impaired at 24 h, 7 days, and 14 days post-exposure. The loss of GABAergic interneurons in the BLA and the decreased interneuron to total number of neurons ratio may be the primary cause of the development of anxiety after nerve agent exposure.
Keywords: Acetylcholinesterase; Anxiety; Basolateral amygdala; Long-term potentiation; Soman; Status epilepticus.
Published by Elsevier Ltd.
Conflict of interest statement
Figures
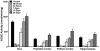





References
-
- Ankarcrona M, Dypbukt JM, Bonfoco E, Zhivotovsky B, Orrenius S, Lipton SA, Nicotera P. Glutamate-induced neuronal death: a succession of necrosis or apoptosis depending on mitochondrial function. Neuron. 1995;15:961–973. - PubMed
-
- Baille V, Clarke PG, Brochier G, Dorandeu F, Verna JM, Four E, Lallement G, Carpentier P. Soman-induced convulsions: the neuropathology revisited. Toxicology. 2005;215:1–24. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

