Mechanisms of vascular dysfunction in mice with endothelium-specific deletion of the PPAR-δ gene
- PMID: 24486511
- PMCID: PMC3962632
- DOI: 10.1152/ajpheart.00761.2013
Mechanisms of vascular dysfunction in mice with endothelium-specific deletion of the PPAR-δ gene
Abstract
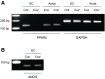
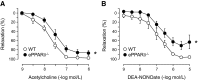
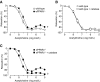
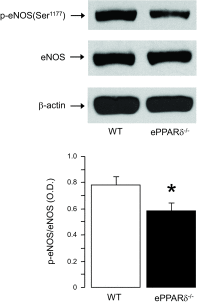
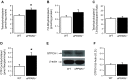
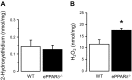
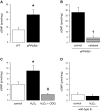
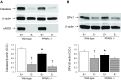
Peroxisome proliferator-activated receptor (PPAR)-δ is a nuclear hormone receptor that is mainly involved in lipid metabolism. Recent studies have suggested that PPAR-δ agonists exert vascular protective effects. The present study was designed to characterize vascular function in mice with genetic inactivation of PPAR-δ in the endothelium. Mice with vascular endothelial cell-specific deletion of the PPAR-δ gene (ePPARδ(-/-) mice) were generated using loxP/Cre technology. ePPARδ(-/-) mice were normotensive and did not display any sign of metabolic syndrome. Endothelium-dependent relaxations to ACh and endothelium-independent relaxations to the nitric oxide (NO) donor diethylammonium (Z)-1-(N,N-diethylamino)diazen-1-ium-1,2-diolate were both significantly impaired in the aorta and carotid arteries of ePPARδ(-/-) mice (P < 0.05). In ePPARδ(-/-) mouse aortas, phosphorylation of endothelial NO synthase at Ser(1177) was significantly decreased (P < 0.05). However, basal levels of cGMP were unexpectedly increased (P < 0.05). Enzymatic activity of GTP-cyclohydrolase I and tetrahydrobiopterin levels were also enhanced in ePPARδ(-/-) mice (P < 0.05). Most notably, endothelium-specific deletion of the PPAR-δ gene significantly decreased protein expressions of catalase and glutathione peroxidase 1 and resulted in increased levels of H2O2 in the aorta (P < 0.05). In contrast, superoxide anion production was unaltered. Moreover, treatment with catalase prevented the endothelial dysfunction and elevation of cGMP detected in aortas of ePPARδ(-/-) mice. The findings suggest that increased levels of cGMP caused by H2O2 impair vasodilator reactivity to endogenous and exogenous NO. We speculate that chronic elevation of H2O2 predisposes PPAR-δ-deficient arteries to oxidative stress and vascular dysfunction.
Keywords: carotid artery; endothelial dysfunction; endothelial nitric oxide synthase; hydrogen peroxide; nitric oxide; peroxisome proliferator-activated receptor-δ; tetrahydrobiopterin.
Figures


Similar articles
-
Activation of PPARδ prevents endothelial dysfunction induced by overexpression of amyloid-β precursor protein.Cardiovasc Res. 2012 Dec 1;96(3):504-12. doi: 10.1093/cvr/cvs266. Epub 2012 Aug 10. Cardiovasc Res. 2012. PMID: 22886847 Free PMC article.
-
Endothelial PPAR-γ provides vascular protection from IL-1β-induced oxidative stress.Am J Physiol Heart Circ Physiol. 2016 Jan 1;310(1):H39-48. doi: 10.1152/ajpheart.00490.2015. Epub 2015 Nov 13. Am J Physiol Heart Circ Physiol. 2016. PMID: 26566726 Free PMC article.
-
Hypercholesterolemia impairs endothelium-dependent relaxations in common carotid arteries of apolipoprotein e-deficient mice.Stroke. 2001 Nov;32(11):2658-64. doi: 10.1161/hs1101.097393. Stroke. 2001. PMID: 11692031
-
Endothelial dysfunction, endothelial nitric oxide bioavailability, tetrahydrobiopterin, and 5-methyltetrahydrofolate in cardiovascular disease. Where are we with therapy?Microvasc Res. 2018 Sep;119:7-12. doi: 10.1016/j.mvr.2018.03.012. Epub 2018 Mar 27. Microvasc Res. 2018. PMID: 29596860 Review.
-
Tetrahydrobiopterin: a vascular redox target to improve endothelial function.Curr Vasc Pharmacol. 2012 Nov;10(6):705-8. doi: 10.2174/157016112803520819. Curr Vasc Pharmacol. 2012. PMID: 23259560 Free PMC article. Review.
Cited by
-
The mitochondrial thioredoxin reductase system (TrxR2) in vascular endothelium controls peroxynitrite levels and tissue integrity.Proc Natl Acad Sci U S A. 2021 Feb 16;118(7):e1921828118. doi: 10.1073/pnas.1921828118. Proc Natl Acad Sci U S A. 2021. PMID: 33579817 Free PMC article.
-
Oleanolic acid ameliorates high glucose-induced endothelial dysfunction via PPARδ activation.Sci Rep. 2017 Jan 9;7:40237. doi: 10.1038/srep40237. Sci Rep. 2017. PMID: 28067284 Free PMC article.
-
Endothelial PPARδ facilitates the post-ischemic vascular repair through interaction with HIF1α.Theranostics. 2022 Jan 24;12(4):1855-1869. doi: 10.7150/thno.69017. eCollection 2022. Theranostics. 2022. PMID: 35198077 Free PMC article.
-
Myosin light chain kinase inhibitor ML7 improves vascular endothelial dysfunction via tight junction regulation in a rabbit model of atherosclerosis.Mol Med Rep. 2015 Sep;12(3):4109-4116. doi: 10.3892/mmr.2015.3973. Epub 2015 Jun 22. Mol Med Rep. 2015. PMID: 26096176 Free PMC article.
-
Inactivation of BACE1 increases expression of endothelial nitric oxide synthase in cerebrovascular endothelium.J Cereb Blood Flow Metab. 2022 Oct;42(10):1920-1932. doi: 10.1177/0271678X221105683. Epub 2022 Jun 8. J Cereb Blood Flow Metab. 2022. PMID: 35673977 Free PMC article.
References
-
- Aebi H. Catalase in vitro. Methods Enzymol 105: 121–126, 1984 - PubMed
-
- Ali F, Ali NS, Bauer A, Boyle JJ, Hamdulay SS, Haskard DO, Randi AM, Mason JC. PPARδ and PGC1α act cooperatively to induce haem oxygenase-1 and enhance vascular endothelial cell resistance to stress. Cardiovasc Res 85: 701–710, 2010 - PubMed
-
- Atochin DN, Wang A, Liu VW, Critchlow JD, Dantas AP, Looft-Wilson R, Murata T, Salomone S, Shin HK, Ayata C, Moskowitz MA, Michel T, Sessa WC, Huang PL. The phosphorylation state of eNOS modulates vascular reactivity and outcome of cerebral ischemia in vivo. J Clin Invest 117: 1961–1967, 2007 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL105278/HL/NHLBI NIH HHS/United States
- P42 ES010337/ES/NIEHS NIH HHS/United States
- HL-088093/HL/NHLBI NIH HHS/United States
- HHMI/Howard Hughes Medical Institute/United States
- R37 DK057978/DK/NIDDK NIH HHS/United States
- HL-91867/HL/NHLBI NIH HHS/United States
- DK-090962/DK/NIDDK NIH HHS/United States
- DK-057978/DK/NIDDK NIH HHS/United States
- P01 HL088093/HL/NHLBI NIH HHS/United States
- R24 DK090962/DK/NIDDK NIH HHS/United States
- HL-105278/HL/NHLBI NIH HHS/United States
- HL-111062/HL/NHLBI NIH HHS/United States
- ES-010337/ES/NIEHS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

