Baseline characteristics and disease burden in patients in the International Paroxysmal Nocturnal Hemoglobinuria Registry
- PMID: 24488565
- PMCID: PMC4008114
- DOI: 10.3324/haematol.2013.093161
Baseline characteristics and disease burden in patients in the International Paroxysmal Nocturnal Hemoglobinuria Registry
Abstract
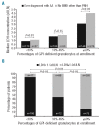
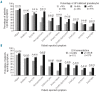
Paroxysmal nocturnal hemoglobinuria is a rare, acquired disease associated with hemolytic anemia, bone marrow failure, thrombosis, and, frequently, poor quality of life. The International PNH Registry is a worldwide, observational, non-interventional study collecting safety, effectiveness, and quality-of-life data from patients with a confirmed paroxysmal nocturnal hemoglobinuria diagnosis or detectable paroxysmal nocturnal hemoglobinuria clone, irrespective of treatment. In addition to evaluating the long-term safety and effectiveness of eculizumab in a global population, the registry aims to improve diagnosis, optimize patient management and outcomes, and enhance the understanding of the natural history of paroxysmal nocturnal hemoglobinuria. Here we report the characteristics of the first 1610 patients enrolled. Median disease duration was 4.6 years. Median granulocyte paroxysmal nocturnal hemoglobinuria clone size was 68.1% (range 0.01-100%). Overall, 16% of patients had a history of thrombotic events and 14% a history of impaired renal function. Therapies included anticoagulation (31%), immunosuppression (19%), and eculizumab (25%). Frequently reported symptoms included fatigue (80%), dyspnea (64%), hemoglobinuria (62%), abdominal pain (44%), and chest pain (33%). Patients suffered from poor quality of life; 23% of patients had been hospitalized due to paroxysmal nocturnal hemoglobinuria-related complications and 17% stated that paroxysmal nocturnal hemoglobinuria was the reason they were not working or were working less. This international registry will provide an ongoing, valuable resource to further the clinical understanding of paroxysmal nocturnal hemoglobinuria.
Figures

References
-
- Hillmen P, Elebute M, Kelly R, Urbano-Ispizua A, Hill A, Rother RP, et al. Long-term effect of the complement inhibitor eculizumab on kidney function in patients with paroxysmal nocturnal hemoglobinuria. Am J Hematol. 2010;85(8):553–9 - PubMed
-
- Kelly RJ, Hill A, Arnold LM, Brooksbank GL, Richards SJ, Cullen M, et al. Long-term treatment with eculizumab in paroxysmal nocturnal hemoglobinuria: sustained efficacy and improved survival. Blood. 2011; 117(25):6786–92 - PubMed
-
- Hillmen P, Lewis SM, Bessler M, Luzzatto L, Dacie JV. Natural history of paroxysmal nocturnal hemoglobinuria. N Engl J Med. 1995;333(19):1253–8 - PubMed
-
- Socié G, Mary JY, de Gramont A, Rio B, Leporrier M, Rose C, et al. Paroxysmal nocturnal haemoglobinuria: long-term follow-up and prognostic factors. French Society of Haematology. Lancet. 1996;348(9027):573–7 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

