Aging of blood can be tracked by DNA methylation changes at just three CpG sites
- PMID: 24490752
- PMCID: PMC4053864
- DOI: 10.1186/gb-2014-15-2-r24
Aging of blood can be tracked by DNA methylation changes at just three CpG sites
Abstract
Background: Human aging is associated with DNA methylation changes at specific sites in the genome. These epigenetic modifications may be used to track donor age for forensic analysis or to estimate biological age.
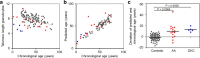
Results: We perform a comprehensive analysis of methylation profiles to narrow down 102 age-related CpG sites in blood. We demonstrate that most of these age-associated methylation changes are reversed in induced pluripotent stem cells (iPSCs). Methylation levels at three age-related CpGs--located in the genes ITGA2B, ASPA and PDE4C--were subsequently analyzed by bisulfite pyrosequencing of 151 blood samples. This epigenetic aging signature facilitates age predictions with a mean absolute deviation from chronological age of less than 5 years. This precision is higher than age predictions based on telomere length. Variation of age predictions correlates moderately with clinical and lifestyle parameters supporting the notion that age-associated methylation changes are associated more with biological age than with chronological age. Furthermore, patients with acquired aplastic anemia or dyskeratosis congenita--two diseases associated with progressive bone marrow failure and severe telomere attrition--are predicted to be prematurely aged.
Conclusions: Our epigenetic aging signature provides a simple biomarker to estimate the state of aging in blood. Age-associated DNA methylation changes are counteracted in iPSCs. On the other hand, over-estimation of chronological age in bone marrow failure syndromes is indicative for exhaustion of the hematopoietic cell pool. Thus, epigenetic changes upon aging seem to reflect biological aging of blood.
Figures


References
-
- Song Z, Von FG, Liu Y, Kraus JM, Torrice C, Dillon P, Rudolph-Watabe M, Ju Z, Kestler HA, Sanoff H, Lenhard RK. Lifestyle impacts on the aging-associated expression of biomarkers of DNA damage and telomere dysfunction in human blood. Aging Cell. 2010;9:607–615. doi: 10.1111/j.1474-9726.2010.00583.x. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

