Contributions of signaling by dopamine neurons in dorsal striatum to cognitive behaviors corresponding to those observed in Parkinson's disease
- PMID: 24491966
- PMCID: PMC4001780
- DOI: 10.1016/j.nbd.2014.01.017
Contributions of signaling by dopamine neurons in dorsal striatum to cognitive behaviors corresponding to those observed in Parkinson's disease
Abstract
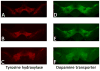
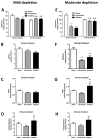
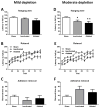
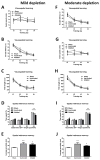
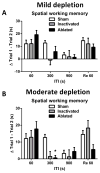
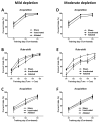
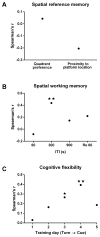
Although the cardinal features of Parkinson's disease (PD) are motor symptoms, PD also causes cognitive deficits including cognitive flexibility and working memory, which are strongly associated with prefrontal cortex (PFC) functions. Yet, early stage PD is not characterized by pathology in the PFC but by a loss of dopaminergic (DA) projections from the substantia nigra to the dorsal striatum. Moreover, the degree to which PD symptoms can be ascribed to the loss of DA alone or to the loss of DA neurons is unknown. We addressed these issues by comparing mouse models of either chronic DA depletion or loss of DA projections to the dorsal striatum. We achieved equal levels of striatal DA reduction in both models which ranged from mild (~25%) to moderate (~60%). Both models displayed DA concentration-dependent reductions of motor function as well as mild deficits of cognitive flexibility and working memory. Interestingly, whereas both motor function and cognitive flexibility were more severely impaired after mild ablation of DA neurons as compared to mild loss of DA alone, both models had equal deficits after moderate loss of DA. Our results confirm contributions of nigro-striatal dopamine signaling to cognitive behaviors that are affected in early stage PD. Furthermore, our findings suggest that the phenotype after ablation of DA neurons accrues from factors beyond the mere loss of DA.
Keywords: 6-Hydroxydopamine; Dopamine-deficient mice; Executive function; Neurodegeneration; Tyrosine hydroxylase; Viral inactivation.
Published by Elsevier Inc.
Figures
Similar articles
-
Relative contributions of severe dopaminergic neuron ablation and dopamine depletion to cognitive impairment.Exp Neurol. 2015 Sep;271:205-14. doi: 10.1016/j.expneurol.2015.06.013. Epub 2015 Jun 14. Exp Neurol. 2015. PMID: 26079646 Free PMC article.
-
RGS Proteins as Critical Regulators of Motor Function and Their Implications in Parkinson's Disease.Mol Pharmacol. 2020 Dec;98(6):730-738. doi: 10.1124/mol.119.118836. Epub 2020 Feb 3. Mol Pharmacol. 2020. PMID: 32015009 Free PMC article. Review.
-
Pitx3 deficiency produces decreased dopamine signaling and induces motor deficits in Pitx3(-/-) mice.Neurobiol Aging. 2015 Dec;36(12):3314-3320. doi: 10.1016/j.neurobiolaging.2015.08.012. Epub 2015 Aug 20. Neurobiol Aging. 2015. PMID: 26363812 Free PMC article.
-
Contributions of striatal dopamine signaling to the modulation of cognitive flexibility.Biol Psychiatry. 2011 Apr 1;69(7):704-7. doi: 10.1016/j.biopsych.2010.09.033. Epub 2010 Nov 11. Biol Psychiatry. 2011. PMID: 21074144 Free PMC article.
-
Differential striatal spine pathology in Parkinson's disease and cocaine addiction: a key role of dopamine?Neuroscience. 2013 Oct 22;251:2-20. doi: 10.1016/j.neuroscience.2013.07.011. Epub 2013 Jul 16. Neuroscience. 2013. PMID: 23867772 Free PMC article. Review.
Cited by
-
Loss of glutamate signaling from the thalamus to dorsal striatum impairs motor function and slows the execution of learned behaviors.NPJ Parkinsons Dis. 2018 Aug 2;4:23. doi: 10.1038/s41531-018-0060-6. eCollection 2018. NPJ Parkinsons Dis. 2018. PMID: 30083593 Free PMC article.
-
Dopamine Deficiency Reduces Striatal Cholinergic Interneuron Function in Models of Parkinson's Disease.Neuron. 2019 Sep 25;103(6):1056-1072.e6. doi: 10.1016/j.neuron.2019.06.013. Epub 2019 Jul 16. Neuron. 2019. PMID: 31324539 Free PMC article.
-
Neuropsychiatric and Cognitive Deficits in Parkinson's Disease and Their Modeling in Rodents.Biomedicines. 2021 Jun 17;9(6):684. doi: 10.3390/biomedicines9060684. Biomedicines. 2021. PMID: 34204380 Free PMC article. Review.
-
Partial lesion of the nigrostriatal dopamine pathway in rats impairs egocentric learning but not spatial learning or behavioral flexibility.Behav Neurosci. 2017 Apr;131(2):135-142. doi: 10.1037/bne0000189. Epub 2017 Feb 20. Behav Neurosci. 2017. PMID: 28221082 Free PMC article.
-
Synergy of Distinct Dopamine Projection Populations in Behavioral Reinforcement.Neuron. 2020 Mar 4;105(5):909-920.e5. doi: 10.1016/j.neuron.2019.11.024. Epub 2019 Dec 23. Neuron. 2020. PMID: 31879163 Free PMC article.
References
-
- Aarsland D, et al. Cognitive impairment in incident, untreated Parkinson disease: the Norwegian ParkWest study. Neurology. 2009;72:1121–6. - PubMed
-
- Ahlskog JE. Beating a dead horse: dopamine and Parkinson disease. Neurology. 2007;69:1701–11. - PubMed
-
- Antonini A, et al. A reassessment of risks and benefits of dopamine agonists in Parkinson’s disease. Lancet Neurol. 2009;8:929–37. - PubMed
-
- Baunez C, Robbins TW. Effects of dopamine depletion of the dorsal striatum and further interaction with subthalamic nucleus lesions in an attentional task in the rat. Neuroscience. 1999;92:1343–56. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

