Dronedarone, an amiodarone analog with improved anti-Leishmania mexicana efficacy
- PMID: 24492373
- PMCID: PMC4023776
- DOI: 10.1128/AAC.01240-13
Dronedarone, an amiodarone analog with improved anti-Leishmania mexicana efficacy
Abstract
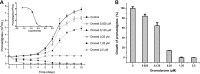
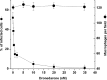
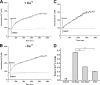
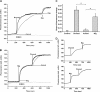
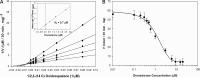
Dronedarone and amiodarone are cationic lipophilic benzofurans used to treat cardiac arrhythmias. They also have activity against the parasitic protozoan Trypanosoma cruzi, the causative agent of Chagas' disease. They function by disrupting intracellular Ca2+ homeostasis of the parasite and by inhibiting membrane sterol (ergosterol) biosynthesis. Amiodarone also has activity against Leishmania mexicana, suggesting that dronedarone might likewise be active against this organism. This might be of therapeutic interest, since dronedarone is thought to have fewer side effects in humans than does amiodarone. We show here that dronedarone effectively inhibits the growth of L. mexicana promastigotes in culture and, more importantly, has excellent activity against amastigotes inside infected macrophages (the clinically relevant form) without affecting the host cell, with the 50% inhibitory concentrations against amastigotes being 3 orders of magnitude lower than those obtained previously with T. cruzi amastigotes (0.65 nM versus 0.75 μM). As with amiodarone, dronedarone affects intracellular Ca2+ homeostasis in the parasite, inducing an elevation of intracellular Ca2+ levels. This is achieved by rapidly collapsing the mitochondrial membrane potential and inducing an alkalinization of acidocalcisomes at a rate that is faster than that observed with amiodarone. We also show that dronedarone inhibits parasite oxidosqualene cyclase, a key enzyme in ergosterol biosynthesis known to be vital for survival. Overall, our results suggest the possibility of repurposing dronedarone as a treatment for cutaneous, and perhaps other, leishmaniases.
Figures



References
-
- Benaim G, Sanders J, Garcia-Marchan Y, Colina C, Lira R, Caldera A, Payares G, Sanoja C, Burgos J, Leon-Rossell A, Concepcion J, Schijman A, Levin M, Oldfield E, Urbina J. 2006. Amiodarone has intrinsic anti-Trypanosoma cruzi activity and acts synergistically with posaconazole. J. Med. Chem. 49:892–899. 10.1021/jm050691f - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

