Angiographic validation of the American College of Cardiology Foundation-the Society of Thoracic Surgeons Collaboration on the Comparative Effectiveness of Revascularization Strategies study
- PMID: 24496239
- PMCID: PMC5656245
- DOI: 10.1161/CIRCINTERVENTIONS.113.000679
Angiographic validation of the American College of Cardiology Foundation-the Society of Thoracic Surgeons Collaboration on the Comparative Effectiveness of Revascularization Strategies study
Abstract
Background: The goal of this study was to compare angiographic interpretation of coronary arteriograms by sites in community practice versus those made by a centralized angiographic core laboratory.
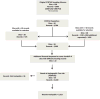
Methods and results: The study population consisted of 2013 American College of Cardiology-National Cardiovascular Data Registry (ACC-NCDR) records with 2- and 3- vessel coronary disease from 54 sites in 2004 to 2007. The primary analysis compared Registry (NCDR)-defined 2- and 3-vessel disease versus those from an angiographic core laboratory analysis. Vessel-level kappa coefficients suggested moderate agreement between NCDR and core laboratory analysis, ranging from kappa=0.39 (95% confidence intervals, 0.32-0.45) for the left anterior descending artery to kappa=0.59 (95% confidence intervals, 0.55-0.64) for the right coronary artery. Overall, 6.3% (n=127 out of 2013) of those patients identified with multivessel disease at NCDR sites had had 0- or 1-vessel disease by core laboratory reading. There was no directional bias with regard to overcall, that is, 12.3% of cases read as 3-vessel disease by the sites were read as <3-vessel disease by the core laboratory, and 13.9% of core laboratory 3-vessel cases were read as <3-vessel by the sites. For a subset of patients with left main coronary disease, registry overcall was not linked to increased rates of mortality or myocardial infarction.
Conclusions: There was only modest agreement between angiographic readings in clinical practice and those from an independent core laboratory. Further study will be needed because the implications for patient management are uncertain.
Keywords: angiography; catheterization; percutaneous coronary intervention; program evaluation.
Conflict of interest statement
The primary author reports no conflicts. Dr Peterson has received consulting fees and honoraria from Janssen Pharmaceuticals, Inc, Genentech, Boehringer Ingelheim as well as research grants from Eli Lilly and Janssen Pharmaceuticals, Inc. Dr Weintraub has received consulting fees and honoraria from Eli Lilly, Bristol Myers Squibb, Pfizer, and Amarin. Dr Gibson has received consulting fees and honoraria from Johnson & Johnson Corp, Bayer Corporation, Ischemix, Inc, BCRI, Sanofi-Aventis Corp, Genentech, Inc, Merck & Co, CSL Behring, Biogen Idec, Daiichi Sankyo, Inc, Bristol Meyer Squibb, The Medicines Company, Janssen Pharmaceuticals, Inc, Regado Biosciences, Inc, St. Jude Medical Corp., Ortho McNeil, Portola Pharmaceuticals, Eli Lilly, and GlaxoSmithKline, as well as research grants from Lantheus Medical Imaging, Janssen Pharmaceuticals, Inc, Bayer Corporation, Genentech Inc, Merck & Co, Atrium Medical Systems, Roche Diagnostics, Ikaria, Inc, Portola Pharmaceuticals, Johnson & Johnson Corp, Angel Medical Corporation, Volcano Corp, Stealth Peptides, Sanofi-Aventis, Walk Vascular, and St Jude’s Medical, as well as other financial support from UpToDate in Cardiovascular Medicine. All other authors report no conflicts.
Figures

Similar articles
-
ACCF/SCAI/STS/AATS/AHA/ASNC 2009 Appropriateness Criteria for Coronary Revascularization: A Report of the American College of Cardiology Foundation Appropriateness Criteria Task Force, Society for Cardiovascular Angiography and Interventions, Society of Thoracic Surgeons, American Association for Thoracic Surgery, American Heart Association, and the American Society of Nuclear Cardiology: Endorsed by the American Society of Echocardiography, the Heart Failure Society of America, and the Society of Cardiovascular Computed Tomography.Circulation. 2009 Mar 10;119(9):1330-52. doi: 10.1161/CIRCULATIONAHA.108.191768. Epub 2009 Jan 8. Circulation. 2009. PMID: 19131581
-
ASCERT: the American College of Cardiology Foundation--the Society of Thoracic Surgeons Collaboration on the comparative effectiveness of revascularization strategies.JACC Cardiovasc Interv. 2010 Jan;3(1):124-6. doi: 10.1016/j.jcin.2009.11.005. JACC Cardiovasc Interv. 2010. PMID: 20129582 Free PMC article. No abstract available.
-
ACCF/SCAI/STS/AATS/AHA/ASNC/HFSA/SCCT 2012 Appropriate use criteria for coronary revascularization focused update: a report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, Society for Cardiovascular Angiography and Interventions, Society of Thoracic Surgeons, American Association for Thoracic Surgery, American Heart Association, American Society of Nuclear Cardiology, and the Society of Cardiovascular Computed Tomography.J Am Coll Cardiol. 2012 Feb 28;59(9):857-81. doi: 10.1016/j.jacc.2011.12.001. Epub 2012 Jan 30. J Am Coll Cardiol. 2012. PMID: 22296741
-
ACCF/SCAI/AATS/AHA/ASE/ASNC/HFSA/HRS/SCCM/SCCT/SCMR/STS 2012 appropriate use criteria for diagnostic catheterization: American College of Cardiology Foundation Appropriate Use Criteria Task Force Society for Cardiovascular Angiography and Interventions American Association for Thoracic Surgery American Heart Association, American Society of Echocardiography American Society of Nuclear Cardiology Heart Failure Society of America Heart Rhythm Society, Society of Critical Care Medicine Society of Cardiovascular Computed Tomography Society for Cardiovascular Magnetic Resonance Society of Thoracic Surgeons.Catheter Cardiovasc Interv. 2012 Sep 1;80(3):E50-81. doi: 10.1002/ccd.24467. Epub 2012 Jun 7. Catheter Cardiovasc Interv. 2012. PMID: 22678595 Review.
-
ACC/AATS/AHA/ASE/ASNC/SCAI/SCCT/STS 2017 Appropriate Use Criteria for Coronary Revascularization in Patients With Stable Ischemic Heart Disease: A Report of the American College of Cardiology Appropriate Use Criteria Task Force, American Association for Thoracic Surgery, American Heart Association, American Society of Echocardiography, American Society of Nuclear Cardiology, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, and Society of Thoracic Surgeons.J Am Coll Cardiol. 2017 May 2;69(17):2212-2241. doi: 10.1016/j.jacc.2017.02.001. Epub 2017 Mar 10. J Am Coll Cardiol. 2017. PMID: 28291663 Review. No abstract available.
Cited by
-
Optimizing ensemble U-Net architectures for robust coronary vessel segmentation in angiographic images.Sci Rep. 2024 Mar 19;14(1):6640. doi: 10.1038/s41598-024-57198-5. Sci Rep. 2024. PMID: 38503839 Free PMC article.
-
Meta-analysis and systematic review of intravascular ultrasound versus angiography-guided drug eluting stent implantation in left main coronary disease in 4592 patients.BMC Cardiovasc Disord. 2018 Jun 14;18(1):115. doi: 10.1186/s12872-018-0843-z. BMC Cardiovasc Disord. 2018. PMID: 29898668 Free PMC article.
-
Central Core Laboratory versus Site Interpretation of Coronary CT Angiography: Agreement and Association with Cardiovascular Events in the PROMISE Trial.Radiology. 2018 Apr;287(1):87-95. doi: 10.1148/radiol.2017172181. Epub 2017 Nov 27. Radiology. 2018. PMID: 29178815 Free PMC article. Clinical Trial.
References
-
- Costa RA, Reiber JH. QCA editorial. Int J Cardiovasc Imaging. 2011;27:155–156. - PubMed
-
- Brener SJ, Haq SA, Bose S, Sacchi TJ. Three-year survival after percutaneous coronary intervention according to appropriateness criteria for revascularization. J Invasive Cardiol. 2009;21:554–557. - PubMed
-
- Stone GW, Moses JW. Interventional cardiology: how should the appropriateness of PCI be judged? Nat Rev Cardiol. 2011;8:544–546. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

