B-CLL cells acquire APC- and CTL-like phenotypic characteristics after stimulation with CpG ODN and IL-21
- PMID: 24497611
- PMCID: PMC4133571
- DOI: 10.1093/intimm/dxu001
B-CLL cells acquire APC- and CTL-like phenotypic characteristics after stimulation with CpG ODN and IL-21
Abstract
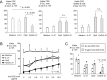
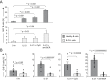
CpG oligodeoxynucleotides (CpG) and IL-21 are two promising agents for the treatment of B-cell chronic lymphocytic leukemia (B-CLL). Recently, we reported that the combination of CpG and IL-21 (CpG/IL-21) can induce granzyme B (GrB)-dependent apoptosis in B-CLL cells. Here, we demonstrate that treatment of B-CLL cells with CpG and IL-21 results in the development of antigen-presenting cell (APC)-like cells with cytotoxic features. These properties eventually give rise to B-CLL cell apoptosis, independently of their cytogenetic phenotype, whereas normal B-cell survival is not negatively affected by CpG/IL-21. APC- and CTL-typical molecules found to be up-regulated in CpG/IL-21-stimulated B-CLL cells include GrB, perforin, T-bet, monokine-induced by IFN-γ and IFN-γ-inducible protein 10 (IP-10), as well as molecules important for cell adhesion, antigen cross-presentation and costimulation. Also induced are molecules involved in GrB induction, trafficking and processing, whereas the GrB inhibitor Serpin B9 [formerly proteinase inhibitor-9 (PI-9)] is down-modulated by CpG/IL-21. In conclusion, CpG/IL-21-stimulated B-CLL cells acquire features that are reminiscent of killer dendritic cells, and which result in enhanced immunogenicity, cytotoxicity and apoptosis. Our results provide novel insights into the aberrant immune state of B-CLL cells and may establish a basis for the development of an innovative cellular vaccination approach in B-CLL.
Keywords: cross-presentation; cytotoxic B cells; immunogenization; leukemia vaccine; toll-like receptor agonist.
© The Japanese Society for Immunology. 2014. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures






Similar articles
-
T-bet suppresses proliferation of malignant B cells in chronic lymphocytic leukemia.Blood. 2024 Aug 1;144(5):510-524. doi: 10.1182/blood.2023021990. Blood. 2024. PMID: 38684038 Free PMC article.
-
[Effect of type 2 innate lymphocytes on Treg and CD8+ T cell function through IL-9 in chronic lymphocytic leukemia].Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2025 Aug;41(8):673-679. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2025. PMID: 40670130 Chinese.
-
B-chronic lymphocytic leukemia cells and other B cells can produce granzyme B and gain cytotoxic potential after interleukin-21-based activation.Blood. 2006 Oct 15;108(8):2712-9. doi: 10.1182/blood-2006-03-014001. Epub 2006 Jun 29. Blood. 2006. PMID: 16809616 Free PMC article.
-
Systemic treatments for metastatic cutaneous melanoma.Cochrane Database Syst Rev. 2018 Feb 6;2(2):CD011123. doi: 10.1002/14651858.CD011123.pub2. Cochrane Database Syst Rev. 2018. PMID: 29405038 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
Cited by
-
CpG oligodeoxynucleotide CpG-685 upregulates functional interleukin-21 receptor on chronic lymphocytic leukemia B cells through an NF-κB mediated pathway.Oncotarget. 2015 Jun 30;6(18):15931-9. doi: 10.18632/oncotarget.3285. Oncotarget. 2015. PMID: 26158860 Free PMC article.
-
Regulatory B Cells-Immunopathological and Prognostic Potential in Humans.Cells. 2024 Feb 18;13(4):357. doi: 10.3390/cells13040357. Cells. 2024. PMID: 38391970 Free PMC article. Review.
-
GIFT4 fusokine converts leukemic B cells into immune helper cells.J Transl Med. 2016 Apr 27;14(1):106. doi: 10.1186/s12967-016-0865-1. J Transl Med. 2016. PMID: 27118475 Free PMC article.
-
The Biological Effects of IL-21 Signaling on B-Cell-Mediated Responses in Organ Transplantation.Front Immunol. 2016 Aug 23;7:319. doi: 10.3389/fimmu.2016.00319. eCollection 2016. Front Immunol. 2016. PMID: 27602031 Free PMC article. Review.
-
IL-21: a pleiotropic cytokine with potential applications in oncology.J Immunol Res. 2015;2015:696578. doi: 10.1155/2015/696578. Epub 2015 Apr 15. J Immunol Res. 2015. PMID: 25961061 Free PMC article. Review.
References
-
- O’Brien S., del Giglio A., Keating M. 1995. Advances in the biology and treatment of B-cell chronic lymphocytic leukemia. Blood 85:307. - PubMed
-
- Jahrsdörfer B., Wooldridge J. E., Blackwell S. E., Taylor C. M., Link B. K., Weiner G. J. 2005. Good prognosis cytogenetics in B-cell chronic lymphocytic leukemia is associated in vitro with low susceptibility to apoptosis and enhanced immunogenicity. Leukemia 19:759. - PubMed
-
- Longo P. G., Laurenti L., Gobessi S., et al. 2007. The Akt signaling pathway determines the different proliferative capacity of chronic lymphocytic leukemia B-cells from patients with progressive and stable disease. Leukemia 21:110. - PubMed
-
- Rezvany M. R., Jeddi-Tehrani M., Rabbani H., et al. 2000. Autologous T lymphocytes may specifically recognize leukaemic B cells in patients with chronic lymphocytic leukaemia. Br. J. Haematol. 111:608. - PubMed
-
- Kollgaard T., Petersen S. L., Hadrup S. R., et al. 2005. Evidence for involvement of clonally expanded CD8+ T cells in anticancer immune responses in CLL patients following nonmyeloablative conditioning and hematopoietic cell transplantation. Leukemia 19:2273. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

