Aggregation gatekeeper and controlled assembly of Trpzip β-hairpins
- PMID: 24498924
- PMCID: PMC3985754
- DOI: 10.1021/bi401568a
Aggregation gatekeeper and controlled assembly of Trpzip β-hairpins
Abstract
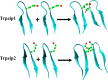
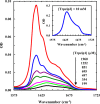
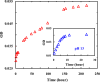
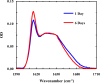
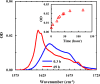
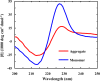
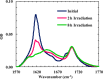
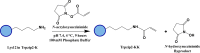
Protein and peptide aggregation is an important issue both in vivo and in vitro. Herein, we examine the aggregation behaviors of two well-studied β-hairpins, Trpzip1 and Trpzip2. Previous studies suggested that Trpzip2 remains monomeric up to a concentration of ~15 mM whereas Trpzip1 readily aggregates at micromolar concentrations at acidic or neutral pH. This disparity is puzzling considering that these two peptides differ only in their turn sequences (i.e., GN vs NG). We hypothesize that these peptides can aggregate from their folded states via native edge-to-edge interactions and that the Lys8 residue in Trpzip2 is a more effective aggregation gatekeeper, because of a more favorable orientation. In support of this hypothesis, we find that increasing the pH to 13 or replacing Lys8 with a hydrophobic and photolabile Lys analogue, Lys(nvoc), leads to a significant increase in the aggregation propensity of Trpzip2, and that the aggregation of this Trpzip2 mutant can be reversed upon restoring the native Lys side chain via photocleavage of the nvoc moiety. In addition, we find that while both Trpzip1 and Trpzip2 form parallel β-sheet aggregates, the Lys(nvoc) Trpzip2 mutant forms antiparallel β-sheets and more stable fibrils. Taken together, these findings provide another example showing how sensitive peptide and protein aggregation is to minor sequence variation and that it is possible to use a photolabile non-natural amino acid, such as Lys(nvoc), to tune the rate of peptide aggregation and to control fibrillar structure.
Figures
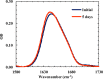
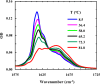
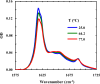


Similar articles
-
Folding thermodynamics of β-hairpins studied by replica-exchange molecular dynamics simulations.Proteins. 2015 Jul;83(7):1307-15. doi: 10.1002/prot.24827. Epub 2015 May 29. Proteins. 2015. PMID: 25973961
-
Light-triggered disassembly of amyloid fibrils.Langmuir. 2012 Aug 28;28(34):12588-92. doi: 10.1021/la302626d. Epub 2012 Aug 16. Langmuir. 2012. PMID: 22867440 Free PMC article.
-
Role of different β-turns in β-hairpin conformation and stability studied by optical spectroscopy.Proteins. 2012 Jan;80(1):44-60. doi: 10.1002/prot.23140. Epub 2011 Oct 12. Proteins. 2012. PMID: 21989967
-
Folding mechanism of β-hairpin trpzip2: heterogeneity, transition state and folding pathways.Int J Mol Sci. 2009 Jun 22;10(6):2838-2848. doi: 10.3390/ijms10062838. Int J Mol Sci. 2009. PMID: 19582232 Free PMC article. Review.
-
Elucidating the Structures of Amyloid Oligomers with Macrocyclic β-Hairpin Peptides: Insights into Alzheimer's Disease and Other Amyloid Diseases.Acc Chem Res. 2018 Mar 20;51(3):706-718. doi: 10.1021/acs.accounts.7b00554. Epub 2018 Mar 6. Acc Chem Res. 2018. PMID: 29508987 Free PMC article. Review.
Cited by
-
Autonomous aggregation suppression by acidic residues explains why chaperones favour basic residues.EMBO J. 2020 Jun 2;39(11):e102864. doi: 10.15252/embj.2019102864. Epub 2020 Apr 1. EMBO J. 2020. PMID: 32237079 Free PMC article.
-
Streamlined Identification of Metallopeptides for Intracellular Catalysis Using Positionally Addressable Combinatorial Libraries.ACS Catal. 2025 May 8;15(10):8624-8632. doi: 10.1021/acscatal.5c00525. eCollection 2025 May 16. ACS Catal. 2025. PMID: 40463646 Free PMC article.
-
Vibrational Approach to the Dynamics and Structure of Protein Amyloids.Molecules. 2019 Jan 6;24(1):186. doi: 10.3390/molecules24010186. Molecules. 2019. PMID: 30621325 Free PMC article. Review.
-
Hierarchical assembly of tryptophan zipper peptides into stress-relaxing bioactive hydrogels.Nat Commun. 2023 Oct 23;14(1):6604. doi: 10.1038/s41467-023-41907-1. Nat Commun. 2023. PMID: 37872151 Free PMC article.
-
N-glycosylation as a eukaryotic protective mechanism against protein aggregation.Sci Adv. 2024 Feb 2;10(5):eadk8173. doi: 10.1126/sciadv.adk8173. Epub 2024 Jan 31. Sci Adv. 2024. PMID: 38295165 Free PMC article.
References
-
- Kelly J. W. (1996) Alternative conformations of amyloidogenic proteins govern their behavior. Curr. Opin. Struct. Biol. 6, 11–17. - PubMed
-
- Dobson C. M. (2003) Protein folding and misfolding. Nature 426, 884–890. - PubMed
-
- Selkoe D. J. (2003) Folding proteins in fatal ways. Nature 426, 900–904. - PubMed
-
- RamirezAlvarado M.; Blanco F. J.; Serrano L. (1996) De novo design and structural analysis of a model β-hairpin peptide system. Nat. Struct. Biol. 3, 604–612. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

