Selectivity for space and time in early areas of the auditory dorsal stream in the rhesus monkey
- PMID: 24501260
- PMCID: PMC4035775
- DOI: 10.1152/jn.00436.2013
Selectivity for space and time in early areas of the auditory dorsal stream in the rhesus monkey
Abstract
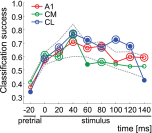
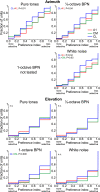
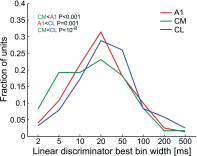
The respective roles of ventral and dorsal cortical processing streams are still under discussion in both vision and audition. We characterized neural responses in the caudal auditory belt cortex, an early dorsal stream region of the macaque. We found fast neural responses with elevated temporal precision as well as neurons selective to sound location. These populations were partly segregated: Neurons in a caudomedial area more precisely followed temporal stimulus structure but were less selective to spatial location. Response latencies in this area were even shorter than in primary auditory cortex. Neurons in a caudolateral area showed higher selectivity for sound source azimuth and elevation, but responses were slower and matching to temporal sound structure was poorer. In contrast to the primary area and other regions studied previously, latencies in the caudal belt neurons were not negatively correlated with best frequency. Our results suggest that two functional substreams may exist within the auditory dorsal stream.
Keywords: azimuth; caudal belt; elevation; latency; temporal precision.
Figures




Similar articles
-
Functional specialization in rhesus monkey auditory cortex.Science. 2001 Apr 13;292(5515):290-3. doi: 10.1126/science.1058911. Science. 2001. PMID: 11303104
-
Effects of stimulus azimuth and intensity on the single-neuron activity in the auditory cortex of the alert macaque monkey.J Neurophysiol. 2006 Dec;96(6):3323-37. doi: 10.1152/jn.00392.2006. Epub 2006 Aug 30. J Neurophysiol. 2006. PMID: 16943318
-
Chronometry on Spike-LFP Responses Reveals the Functional Neural Circuitry of Early Auditory Cortex Underlying Sound Processing and Discrimination.eNeuro. 2018 Jul 3;5(3):ENEURO.0420-17.2018. doi: 10.1523/ENEURO.0420-17.2018. eCollection 2018 May-Jun. eNeuro. 2018. PMID: 29971252 Free PMC article.
-
Processing of complex sounds in the auditory cortex of cat, monkey, and man.Acta Otolaryngol Suppl. 1997;532:34-8. doi: 10.3109/00016489709126142. Acta Otolaryngol Suppl. 1997. PMID: 9442842 Review.
-
Spatial and temporal processing of single auditory cortical neurons and populations of neurons in the macaque monkey.Hear Res. 2011 Jan;271(1-2):115-22. doi: 10.1016/j.heares.2010.03.084. Epub 2010 Apr 27. Hear Res. 2011. PMID: 20430079 Free PMC article. Review.
Cited by
-
Frequency-dependent auditory space representation in the human planum temporale.Front Hum Neurosci. 2014 Jul 22;8:524. doi: 10.3389/fnhum.2014.00524. eCollection 2014. Front Hum Neurosci. 2014. PMID: 25100973 Free PMC article.
-
Where, When, and How: Are they all sensorimotor? Towards a unified view of the dorsal pathway in vision and audition.Cortex. 2018 Jan;98:262-268. doi: 10.1016/j.cortex.2017.10.020. Epub 2017 Nov 3. Cortex. 2018. PMID: 29183630 Free PMC article. Review.
-
Noncanonical Short-Latency Auditory Pathway Directly Activates Deep Cortical Layers.bioRxiv [Preprint]. 2025 Feb 11:2025.01.06.631598. doi: 10.1101/2025.01.06.631598. bioRxiv. 2025. Update in: Nat Commun. 2025 Jul 1;16(1):5911. doi: 10.1038/s41467-025-61020-9. PMID: 39829930 Free PMC article. Updated. Preprint.
-
The evolutionary neuroscience of musical beat perception: the Action Simulation for Auditory Prediction (ASAP) hypothesis.Front Syst Neurosci. 2014 May 13;8:57. doi: 10.3389/fnsys.2014.00057. eCollection 2014. Front Syst Neurosci. 2014. PMID: 24860439 Free PMC article.
-
Widespread and Opponent fMRI Signals Represent Sound Location in Macaque Auditory Cortex.Neuron. 2017 Feb 22;93(4):971-983.e4. doi: 10.1016/j.neuron.2017.01.013. Epub 2017 Feb 9. Neuron. 2017. PMID: 28190642 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

