Caenorhabditis elegans HOPS and CCZ-1 mediate trafficking to lysosome-related organelles independently of RAB-7 and SAND-1
- PMID: 24501423
- PMCID: PMC3967972
- DOI: 10.1091/mbc.E13-09-0521
Caenorhabditis elegans HOPS and CCZ-1 mediate trafficking to lysosome-related organelles independently of RAB-7 and SAND-1
Abstract
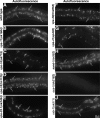
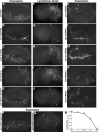
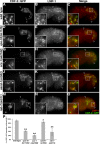
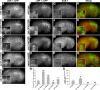
As early endosomes mature, the SAND-1/CCZ-1 complex acts as a guanine nucleotide exchange factor (GEF) for RAB-7 to promote the activity of its effector, HOPS, which facilitates late endosome-lysosome fusion and the consumption of AP-3-containing vesicles. We show that CCZ-1 and the HOPS complex are essential for the biogenesis of gut granules, cell type-specific, lysosome-related organelles (LROs) that coexist with conventional lysosomes in Caenorhabditis elegans intestinal cells. The HOPS subunit VPS-18 promotes the trafficking of gut granule proteins away from lysosomes and functions downstream of or in parallel to the AP-3 adaptor. CCZ-1 also acts independently of AP-3, and ccz-1 mutants mistraffic gut granule proteins. Our results indicate that SAND-1 does not participate in the formation of gut granules. In the absence of RAB-7 activity, gut granules are generated; however, their size and protein composition are subtly altered. These observations suggest that CCZ-1 acts in partnership with a protein other than SAND-1 as a GEF for an alternate Rab to promote gut granule biogenesis. Point mutations in GLO-1, a Rab32/38-related protein, predicted to increase spontaneous guanine nucleotide exchange, specifically suppress the loss of gut granules by ccz-1 and glo-3 mutants. GLO-3 is known to be required for gut granule formation and has homology to SAND-1/Mon1-related proteins, suggesting that CCZ-1 functions with GLO-3 upstream of the GLO-1 Rab, possibly as a GLO-1 GEF. These results support LRO formation occurring via processes similar to conventional lysosome biogenesis, albeit with key molecular differences.
Figures





References
-
- Aoyama M, Sun-Wada GH, Yamamoto A, Yamamoto M, Hamada H, Wada Y. Spatial restriction of bone morphogenetic protein signaling in mouse gastrula through the mVam2-dependent endocytic pathway. Dev Cell. 2012;22:1163–1175. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

