Analysis of synaptic-like microvesicle exocytosis of B-cells using a live imaging technique
- PMID: 24503905
- PMCID: PMC3913683
- DOI: 10.1371/journal.pone.0087758
Analysis of synaptic-like microvesicle exocytosis of B-cells using a live imaging technique
Abstract
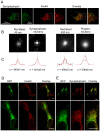
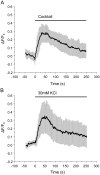
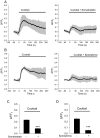
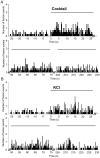
Pancreatic β-cells play central roles in blood glucose homeostasis. Beside insulin, these cells release neurotransmitters and other signaling molecules stored in synaptic-like microvesicles (SLMVs). We monitored SLMV exocytosis by transfecting a synaptophysin-pHluorin construct and by visualizing the cells by Total Internal Reflection Fluorescence (TIRF) microscopy. SLMV fusion was elicited by 20 mM glucose and by depolarizing K(+) concentrations with kinetics comparable to insulin secretion. SLMV exocytosis was prevented by Tetanus and Botulinum-C neurotoxins indicating that the fusion machinery of these organelles includes VAMP-2/-3 and Syntaxin-1, respectively. Sequential visualization of SLMVs by TIRF and epifluorescence microscopy showed that after fusion the vesicle components are rapidly internalized and the organelles re-acidified. Analysis of single fusion episodes revealed the existence of two categories of events. While under basal conditions transient fusion events prevailed, long-lasting episodes were more frequent upon secretagogue exposure. Our observations unveiled similarities between the mechanism of exocytosis of insulin granules and SLMVs. Thus, diabetic conditions characterized by defective insulin secretion are most probably associated also with inappropriate release of molecules stored in SLMVs. The assessment of the contribution of SLMV exocytosis to the manifestation of the disease will be facilitated by the use of the imaging approach described in this study.
Conflict of interest statement
Figures



References
-
- Gromada J, Franklin I, Wollheim CB (2007) Alpha-cells of the endocrine pancreas: 35 years of research but the enigma remains. Endocr Rev 28: 84–116. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

