Key roles for the small leucine-rich proteoglycans in renal and pulmonary pathophysiology
- PMID: 24508120
- PMCID: PMC4074572
- DOI: 10.1016/j.bbagen.2014.01.035
Key roles for the small leucine-rich proteoglycans in renal and pulmonary pathophysiology
Abstract
Background: Small leucine-rich proteoglycans (SLRPs) are molecules that have signaling roles in a multitude of biological processes. In this respect, SLRPs play key roles in the evolution of a variety of diseases throughout the human body.
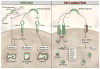
Scope of review: We will critically review current developments in the roles of SLRPs in several types of disease of the kidney and lungs. Particular emphasis will be given to the roles of decorin and biglycan, the best characterized members of the SLRP gene family.
Major conclusions: In both renal and pulmonary disorders, SLRPs are essential elements that regulate several pathophysiological processes including fibrosis, inflammation and tumor progression. Decorin has remarkable antifibrotic and antitumorigenic properties and is considered a valuable potential treatment of these diseases. Biglycan can modulate inflammatory processes in lung and renal inflammation and is a potential target in the treatment of inflammatory conditions.
General significance: SLRPs can serve as either treatment targets or as potential treatment in renal or lung disease. This article is part of a Special Issue entitled Matrix-mediated cell behaviour and properties.
Keywords: Biglycan; Cancer; Decorin; Extracellular matrix; TGF-β; Toll-like receptor.
Copyright © 2014 Elsevier B.V. All rights reserved.
Figures

References
-
- Hocking AM, Shinomura T, McQuillan DJ. Leucine-rich repeat glycoproteins of the extracellular matrix. Matrix Biol. 1998;17:1–19. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

