Differential roles of cardiac and leukocyte derived macrophage migration inhibitory factor in inflammatory responses and cardiac remodelling post myocardial infarction
- PMID: 24508700
- PMCID: PMC4317300
- DOI: 10.1016/j.yjmcc.2014.01.015
Differential roles of cardiac and leukocyte derived macrophage migration inhibitory factor in inflammatory responses and cardiac remodelling post myocardial infarction
Abstract
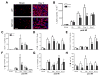
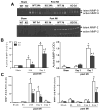
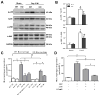
Myocardial infarction (MI) provokes regional inflammation which facilitates the healing, whereas excessive inflammation leads to adverse cardiac remodelling. Our aim was to determine the role of macrophage migration inhibitory factor (MIF) in inflammation and cardiac remodelling following MI. Wild type (WT) or global MIF deficient (MIFKO) mice were subjected to coronary artery occlusion. Compared to WT mice, MIFKO mice had a significantly lower incidence of post-MI cardiac rupture (27% vs. 53%) and amelioration of cardiac remodelling. These were associated with suppressed myocardial leukocyte infiltration, inflammatory mediators' expression, and reduced activity of MMP-2, MMP-9, p38 and JNK MAPK. Infarct myocardium-derived or exogenous MIF mediated macrophage chemotaxis in vitro that was suppressed by inhibition of p38 MAPK or NF-κB. To further dissect the role of MIF derived from different cellular sources in post-MI cardiac remodelling, we generated chimeric mice with MIF deficiency either in bone marrow derived-cells (WT(KO)) or in somatic-cells (KO(WT)). Compared to WT and KO(WT) mice, WT(KO) mice had reduced rupture risk and ameliorated cardiac remodelling, associated with attenuated regional leukocyte infiltration and expression of inflammatory mediators. In contrast, KO(WT) mice had delayed healing and enhanced expression of M1 macrophage markers, but diminished expression of M2 markers during the early healing phase. In conclusion, global MIF deletion protects the heart from post-infarct cardiac rupture and remodelling through suppression of leukocyte infiltration and inflammation. Leukocyte-derived MIF promotes inflammatory responses after MI, whereas cardiac-derived MIF affects early but not ultimate healing process.
Keywords: Healing; Inflammation; Macrophage migration inhibitory factor; Myocardial infarction.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures


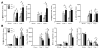
Similar articles
-
Pro-inflammatory action of MIF in acute myocardial infarction via activation of peripheral blood mononuclear cells.PLoS One. 2013 Oct 1;8(10):e76206. doi: 10.1371/journal.pone.0076206. eCollection 2013. PLoS One. 2013. PMID: 24098445 Free PMC article.
-
Cardiomyocyte-specific deficiency of HSPB1 worsens cardiac dysfunction by activating NFκB-mediated leucocyte recruitment after myocardial infarction.Cardiovasc Res. 2019 Jan 1;115(1):154-167. doi: 10.1093/cvr/cvy163. Cardiovasc Res. 2019. PMID: 29982352 Free PMC article.
-
Compartmentalized protective and detrimental effects of endogenous macrophage migration-inhibitory factor mediated by CXCR2 in a mouse model of myocardial ischemia/reperfusion.Arterioscler Thromb Vasc Biol. 2013 Sep;33(9):2180-6. doi: 10.1161/ATVBAHA.113.301633. Epub 2013 Jul 18. Arterioscler Thromb Vasc Biol. 2013. PMID: 23868943 Free PMC article.
-
Cardiac macrophages and their role in ischaemic heart disease.Cardiovasc Res. 2014 May 1;102(2):240-8. doi: 10.1093/cvr/cvu025. Epub 2014 Feb 5. Cardiovasc Res. 2014. PMID: 24501331 Free PMC article. Review.
-
Distinct roles of MIF in the pathogenesis of ischemic heart disease.Cytokine Growth Factor Rev. 2024 Dec;80:121-137. doi: 10.1016/j.cytogfr.2024.10.005. Epub 2024 Oct 16. Cytokine Growth Factor Rev. 2024. PMID: 39438226 Review.
Cited by
-
Regulation of Type 2 Immunity in Myocardial Infarction.Front Immunol. 2019 Jan 29;10:62. doi: 10.3389/fimmu.2019.00062. eCollection 2019. Front Immunol. 2019. PMID: 30761134 Free PMC article. Review.
-
Prototype device for endoventricular beta-emitting radiotracer detection and molecularly-guided intervention.J Nucl Cardiol. 2022 Apr;29(2):663-676. doi: 10.1007/s12350-020-02317-8. Epub 2020 Aug 20. J Nucl Cardiol. 2022. PMID: 32820423 Free PMC article.
-
Matrix Metalloproteinase 2 as a Pharmacological Target in Heart Failure.Pharmaceuticals (Basel). 2022 Jul 25;15(8):920. doi: 10.3390/ph15080920. Pharmaceuticals (Basel). 2022. PMID: 35893744 Free PMC article. Review.
-
Macrophage contribution to the survival of transferred expanded skin flap through angiogenesis.Ann Transl Med. 2023 Mar 31;11(6):248. doi: 10.21037/atm-22-1558. Epub 2023 Jan 30. Ann Transl Med. 2023. PMID: 37082668 Free PMC article.
-
The Role of Cardiokines in Heart Diseases: Beneficial or Detrimental?Biomed Res Int. 2018 Mar 18;2018:8207058. doi: 10.1155/2018/8207058. eCollection 2018. Biomed Res Int. 2018. PMID: 29744364 Free PMC article. Review.
References
-
- Roger VL, Go AS, Lloyd-Jones DM, Benjamin EJ, Berry JD, Borden WB, et al. Executive summary: heart disease and stroke statistics--2012 update: a report from the American Heart Association. Circulation. 2012;125:188–97. - PubMed
-
- Opie LH, Commerford PJ, Gersh BJ, Pfeffer MA. Controversies in ventricular remodelling. Lancet. 2006;367:356–67. - PubMed
-
- Pfeffer MA, Braunwald E. Ventricular remodeling after myocardial infarction. Experimental observations and clinical implications. Circulation. 1990;81:1161–72. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

