A short term quality control tool for biodegradable microspheres
- PMID: 24519488
- PMCID: PMC4037492
- DOI: 10.1208/s12249-013-0052-0
A short term quality control tool for biodegradable microspheres
Abstract
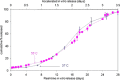
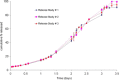
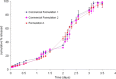
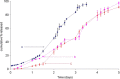
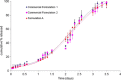
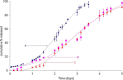
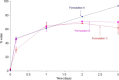
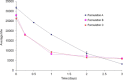
Accelerated in vitro release testing methodology has been developed as an indicator of product performance to be used as a discriminatory quality control (QC) technique for the release of clinical and commercial batches of biodegradable microspheres. While product performance of biodegradable microspheres can be verified by in vivo and/or in vitro experiments, such evaluation can be particularly challenging because of slow polymer degradation, resulting in extended study times, labor, and expense. Three batches of Leuprolide poly(lactic-co-glycolic acid) (PLGA) microspheres having varying morphology (process variants having different particle size and specific surface area) were manufactured by the solvent extraction/evaporation technique. Tests involving in vitro release, polymer degradation and hydration of the microspheres were performed on the three batches at 55°C. In vitro peptide release at 55°C was analyzed using a previously derived modification of the Weibull function termed the modified Weibull equation (MWE). Experimental observations and data analysis confirm excellent reproducibility studies within and between batches of the microsphere formulations demonstrating the predictability of the accelerated experiments at 55°C. The accelerated test method was also successfully able to distinguish the in vitro product performance between the three batches having varying morphology (process variants), indicating that it is a suitable QC tool to discriminate product or process variants in clinical or commercial batches of microspheres. Additionally, data analysis utilized the MWE to further quantify the differences obtained from the accelerated in vitro product performance test between process variants, thereby enhancing the discriminatory power of the accelerated methodology at 55°C.
Figures

Similar articles
-
Effect of processing parameters on characteristics of biodegradable extended-release microspheres containing leuprolide acetate.Drug Dev Ind Pharm. 2024 Dec;50(12):981-994. doi: 10.1080/03639045.2024.2433621. Epub 2024 Dec 1. Drug Dev Ind Pharm. 2024. PMID: 39616598
-
Effect of PLGA raw materials on in vitro and in vivo performance of drug-loaded microspheres.Drug Deliv Transl Res. 2025 Jan;15(1):185-202. doi: 10.1007/s13346-024-01577-y. Epub 2024 Apr 20. Drug Deliv Transl Res. 2025. PMID: 38643259
-
Accelerated in vitro release testing of implantable PLGA microsphere/PVA hydrogel composite coatings.Int J Pharm. 2012 Jan 17;422(1-2):341-8. doi: 10.1016/j.ijpharm.2011.10.020. Epub 2011 Oct 13. Int J Pharm. 2012. PMID: 22016033 Free PMC article.
-
A biodegradable polymeric system for peptide-protein delivery assembled with porous microspheres and nanoparticles, using an adsorption/infiltration process.Int J Nanomedicine. 2013;8:2141-51. doi: 10.2147/IJN.S44482. Epub 2013 Jun 10. Int J Nanomedicine. 2013. PMID: 23788833 Free PMC article.
-
Long-acting PLGA microspheres: Advances in excipient and product analysis toward improved product understanding.Adv Drug Deliv Rev. 2023 Jul;198:114857. doi: 10.1016/j.addr.2023.114857. Epub 2023 May 5. Adv Drug Deliv Rev. 2023. PMID: 37149041 Review.
Cited by
-
Application of a Customised Franz-Type Cell Coupled with HPTLC to Monitor the Timed Release of Bioactive Components in Complex Honey Matrices.Methods Protoc. 2023 Aug 3;6(4):70. doi: 10.3390/mps6040070. Methods Protoc. 2023. PMID: 37623921 Free PMC article.
-
In vitro-in vivo correlation from lactide-co-glycolide polymeric dosage forms.Prog Biomater. 2014 Dec;3(2-4):131-142. doi: 10.1007/s40204-014-0029-4. Epub 2014 Dec 2. Prog Biomater. 2014. PMID: 29470771 Free PMC article.
-
In vitro-in vivo correlation of parenteral risperidone polymeric microspheres.J Control Release. 2015 Nov 28;218:2-12. doi: 10.1016/j.jconrel.2015.09.051. Epub 2015 Sep 28. J Control Release. 2015. PMID: 26423236 Free PMC article.
-
The Effect of Polymer Blends on the In Vitro Release/Degradation and Pharmacokinetics of Moxidectin-Loaded PLGA Microspheres.Int J Mol Sci. 2023 Sep 29;24(19):14729. doi: 10.3390/ijms241914729. Int J Mol Sci. 2023. PMID: 37834176 Free PMC article.
-
PLGA/PLA-Based Long-Acting Injectable Depot Microspheres in Clinical Use: Production and Characterization Overview for Protein/Peptide Delivery.Int J Mol Sci. 2021 Aug 18;22(16):8884. doi: 10.3390/ijms22168884. Int J Mol Sci. 2021. PMID: 34445587 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

