Diversity and plasticity in Rab GTPase nucleotide release mechanism has consequences for Rab activation and inactivation
- PMID: 24520163
- PMCID: PMC3919270
- DOI: 10.7554/eLife.01623
Diversity and plasticity in Rab GTPase nucleotide release mechanism has consequences for Rab activation and inactivation
Abstract
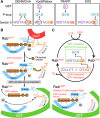
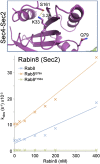
Ras superfamily GTPase activation and inactivation occur by canonical nucleotide exchange and GTP hydrolysis mechanisms. Despite conservation of active-site residues, the Ras-related Rab GTPase activation pathway differs from Ras and between different Rabs. Analysis of DENND1-Rab35, Rabex-Rab5, TRAPP-Rab1 and DrrA-Rab1 suggests Rabs have the potential for activation by distinct GDP-release pathways. Conserved active-site residues in the Rab switch II region stabilising the nucleotide-free form differentiate these pathways. For DENND1-Rab35 and DrrA-Rab1 the Rab active-site glutamine, often mutated to create constitutively active forms, is involved in GEF mediated GDP-release. By contrast, in Rab5 the switch II aspartate is required for Rabex mediated GDP-release. Furthermore, Rab1 switch II glutamine mutants refractory to activation by DrrA can be activated by TRAPP, showing that a single Rab can be activated by more than one mechanistically distinct GDP-release pathway. These findings highlight plasticity in the activation mechanisms of closely related Rab GTPases. DOI: http://dx.doi.org/10.7554/eLife.01623.001.
Keywords: Rab GTPase; membrane traffic; nucleotide exchange factor.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures
Comment in
-
Mutant enzymes challenge all assumptions.Elife. 2014 Feb 11;3:e02171. doi: 10.7554/eLife.02171. Elife. 2014. PMID: 24520166 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

