BRK targets Dok1 for ubiquitin-mediated proteasomal degradation to promote cell proliferation and migration
- PMID: 24523872
- PMCID: PMC3921129
- DOI: 10.1371/journal.pone.0087684
BRK targets Dok1 for ubiquitin-mediated proteasomal degradation to promote cell proliferation and migration
Erratum in
- PLoS One. 2014;9(5):e98814
Abstract
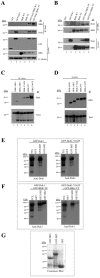
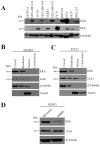
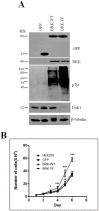
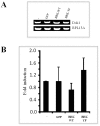
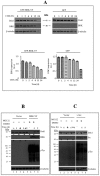
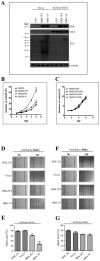
Breast tumor kinase (BRK), also known as protein tyrosine kinase 6 (PTK6), is a non-receptor tyrosine kinase overexpressed in more that 60% of human breast carcinomas. The overexpression of BRK has been shown to sensitize mammary epithelial cells to mitogenic signaling and to promote cell proliferation and tumor formation. The molecular mechanisms of BRK have been unveiled by the identification and characterization of BRK target proteins. Downstream of tyrosine kinases 1 or Dok1 is a scaffolding protein and a substrate of several tyrosine kinases. Herein we show that BRK interacts with and phosphorylates Dok1 specifically on Y362. We demonstrate that this phosphorylation by BRK significantly downregulates Dok1 in a ubiquitin-proteasome-mediated mechanism. Together, these results suggest a novel mechanism of action of BRK in the promotion of tumor formation, which involves the targeting of tumor suppressor Dok1 for degradation through the ubiquitin proteasomal pathway.
Conflict of interest statement
Figures


References
-
- Lee S-K, Shaw AS, Maher SE, Bothwell ALM (1993) p59fyn tyrosine kinase is an essential regulator of p56lck tyrosine kinase activity and early TcR-mediated signalling. - PubMed
-
- Mitchell PJ, Barker KT, Martindale JE, Kamalati T, Lowe PN, et al. (1994) Cloning and characterisation of cDNAs encoding a novel non-receptor tyrosine kinase, brk, expressed in human breast tumours. Oncogene 9: 2383–2390. - PubMed
-
- Barker KT, Jackson LE, Crompton MR (1997) BRK tyrosine kinase expression in a high proportion of human breast carcinomas. Oncogene 15: 799–805. - PubMed
-
- Mitchell PJ, Barker KT, Martindale JE, Kamalati T, Lowe PN, et al. (1994) Cloning and characterization of cDNAs encoding a novel non-receptor tyrosine kinase, brk, expressed in human breast tumours. Oncogene 9: 2383–2390. - PubMed
-
- Ostrander JH, Daniel AR, Lofgren K, Kleer CG, Lange CA (2007) Breast tumor kinase (protein tyrosine kinase 6) regulates heregulin-induced activation of ERK5 and p38 MAP kinases in breast cancer cells. Cancer Res 67: 4199–4209. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

