The R-spondin/Lgr5/Rnf43 module: regulator of Wnt signal strength
- PMID: 24532711
- PMCID: PMC3937510
- DOI: 10.1101/gad.235473.113
The R-spondin/Lgr5/Rnf43 module: regulator of Wnt signal strength
Abstract
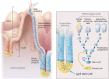
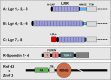
Lgr5 was originally discovered as a common Wnt target gene in adult intestinal crypts and colon cancer. It was subsequently identified as an exquisite marker of multiple Wnt-driven adult stem cell types. Lgr5 and its homologs, Lgr4 and Lgr6, constitute the receptors for R-spondins, potent Wnt signal enhancers and stem cell growth factors. The Lgr5/R-spondin complex acts by neutralizing Rnf43 and Znrf3, two transmembrane E3 ligases that remove Wnt receptors from the stem cell surface. Rnf43/Znrf3 are themselves encoded by Wnt target genes and constitute a negative Wnt feedback loop. Thus, adult stem cells are controlled by an intricate interplay of potent Wnt agonists, antagonists, and anti-antagonists.
Keywords: Lgr5; Rnf43/Znrf3; Rspondin; Wnt signal regulation.
Figures



Similar articles
-
The structural basis of R-spondin recognition by LGR5 and RNF43.Genes Dev. 2013 Jun 15;27(12):1345-50. doi: 10.1101/gad.219915.113. Epub 2013 Jun 11. Genes Dev. 2013. PMID: 23756651 Free PMC article.
-
LGR4 and LGR5 form distinct homodimers that only LGR4 complexes with RNF43/ZNRF3 to provide high affinity binding of R-spondin ligands.Sci Rep. 2023 Jul 4;13(1):10796. doi: 10.1038/s41598-023-37856-w. Sci Rep. 2023. PMID: 37402772 Free PMC article.
-
Interaction with both ZNRF3 and LGR4 is required for the signalling activity of R-spondin.EMBO Rep. 2013 Dec;14(12):1120-6. doi: 10.1038/embor.2013.167. Epub 2013 Oct 29. EMBO Rep. 2013. PMID: 24165923 Free PMC article.
-
The RSPO-LGR4/5-ZNRF3/RNF43 module in liver homeostasis, regeneration, and disease.Hepatology. 2022 Sep;76(3):888-899. doi: 10.1002/hep.32328. Epub 2022 Feb 20. Hepatology. 2022. PMID: 35006616 Review.
-
R-spondins: Multi-mode WNT signaling regulators in adult stem cells.Int J Biochem Cell Biol. 2019 Jan;106:26-34. doi: 10.1016/j.biocel.2018.11.005. Epub 2018 Nov 12. Int J Biochem Cell Biol. 2019. PMID: 30439551 Review.
Cited by
-
New insights in ubiquitin-dependent Wnt receptor regulation in tumorigenesis.In Vitro Cell Dev Biol Anim. 2024 May;60(5):449-465. doi: 10.1007/s11626-024-00855-w. Epub 2024 Feb 21. In Vitro Cell Dev Biol Anim. 2024. PMID: 38383910 Free PMC article. Review.
-
Wnt pathway regulation of intestinal stem cells.J Physiol. 2016 Sep 1;594(17):4837-47. doi: 10.1113/JP271754. Epub 2016 Jun 16. J Physiol. 2016. PMID: 27581568 Free PMC article. Review.
-
Mammary Development and Breast Cancer: A Wnt Perspective.Cancers (Basel). 2016 Jul 13;8(7):65. doi: 10.3390/cancers8070065. Cancers (Basel). 2016. PMID: 27420097 Free PMC article. Review.
-
Validity of pathological diagnosis for early colorectal cancer in genetic background.Cancer Med. 2023 Apr;12(7):8490-8498. doi: 10.1002/cam4.5596. Epub 2023 Feb 3. Cancer Med. 2023. PMID: 36734304 Free PMC article.
-
DACH1 inhibits glioma invasion and tumor growth via the Wnt/catenin pathway.Onco Targets Ther. 2018 Sep 17;11:5853-5863. doi: 10.2147/OTT.S168314. eCollection 2018. Onco Targets Ther. 2018. PMID: 30271168 Free PMC article.
References
-
- Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, et al. 2007. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449: 1003–1007 - PubMed
-
- Barker N, Huch M, Kujala P, van de Wetering M, Snippert HJ, van Es JH, Sato T, Stange DE, Begthel H, van den Born M, et al. 2010. Lgr5(+ve) stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell 6: 25–36 - PubMed
-
- Barker N, Rookmaaker MB, Kujala P, Ng A, Leushacke M, Snippert H, van de Wetering M, Tan S, van Es JH, Huch M et al. 2012. Lgr5(+ve) stem/progenitor cells contribute to nephron formation during kidney development. Cell Reports 2: 540–552 - PubMed
-
- Bell SM, Schreiner CM, Wert SE, Mucenski ML, Scott WJ, Whitsett JA 2008. R-spondin 2 is required for normal laryngeal-tracheal, lung and limb morphogenesis. Development 135: 1049–1058 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
