Developmental and maintenance defects in Rett syndrome neurons identified by a new mouse staging system in vitro
- PMID: 24550777
- PMCID: PMC3914021
- DOI: 10.3389/fncel.2014.00018
Developmental and maintenance defects in Rett syndrome neurons identified by a new mouse staging system in vitro
Abstract
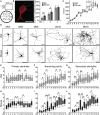
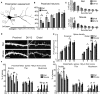
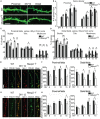
Rett Syndrome (RTT) is a neurodevelopmental disorder associated with intellectual disability, mainly caused by loss-of-function mutations in the MECP2 gene. RTT brains display decreased neuronal size and dendritic arborization possibly caused by either a developmental failure or a deficit in the maintenance of dendritic arbor structure. To distinguish between these two hypotheses, the development of Mecp2-knockout mouse hippocampal neurons was analyzed in vitro. Since a staging system for the in vitro development of mouse neurons was lacking, mouse and rat hippocampal neurons development was compared between 1-15 days in vitro (DIV) leading to a 6-stage model for both species. Mecp2-knockout hippocampal neurons displayed reduced growth of dendritic branches from stage 4 (DIV4) onwards. At stages 5-6 (DIV9-15), synapse number was lowered in Mecp2-knockout neurons, suggesting increased synapse elimination. These results point to both a developmental and a maintenance setback affecting the final shape and function of neurons in RTT.
Keywords: Dendrites; Dendritic Spines; Neuronal morphology; Rett Syndrome; hippocampal neurons; neurodevelopment; staging system.
Figures




References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

