NK cell autoreactivity and autoimmune diseases
- PMID: 24550913
- PMCID: PMC3912987
- DOI: 10.3389/fimmu.2014.00027
NK cell autoreactivity and autoimmune diseases
Abstract
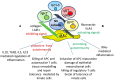
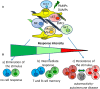
Increasing evidences have pointed out the relevance of natural killer (NK) cells in organ-specific and systemic autoimmune diseases. NK cells bear a plethora of activating and inhibiting receptors that can play a role in regulating reactivity with autologous cells. The activating receptors recognize natural ligands up-regulated on virus-infected or stressed or neoplastic cells. Of note, several autoimmune diseases are thought to be linked to viral infections as one of the first event in inducing autoimmunity. Also, it is conceivable that autoimmunity can be triggered when a dysregulation of innate immunity occurs, activating T and B lymphocytes to react with self-components. This would imply that NK cells can play a regulatory role during adaptive immunity; indeed, innate lymphoid cells (ILCs), comprising the classical CD56(+) NK cells, have a role in maintaining or alternating tissue homeostasis secreting protective and/or pro-inflammatory cytokines. In addition, NK cells display activating receptors involved in natural cytotoxicity and the activating isoforms of receptors for HLA class I that can interact with healthy host cells and induce damage without any evidence of viral infection or neoplastic-induced alteration. In this context, the interrelationship among ILC, extracellular-matrix components, and mesenchymal stromal cells can be considered a key point for the control of homeostasis. Herein, we summarize evidences for a role of NK cells in autoimmune diseases and will give a point of view of the interplay between NK cells and self-cells in triggering autoimmunity.
Keywords: DNAM1; LAIR1; NK cells; NKG2D; autoimmunity; autoreactivity; mesenchymal stromal cells; regulatory NK cells.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

