A modular plasmid assembly kit for multigene expression, gene silencing and silencing rescue in plants
- PMID: 24551083
- PMCID: PMC3923767
- DOI: 10.1371/journal.pone.0088218
A modular plasmid assembly kit for multigene expression, gene silencing and silencing rescue in plants
Abstract
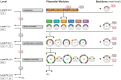
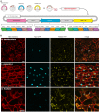
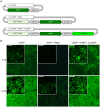
The Golden Gate (GG) modular assembly approach offers a standardized, inexpensive and reliable way to ligate multiple DNA fragments in a pre-defined order in a single-tube reaction. We developed a GG based toolkit for the flexible construction of binary plasmids for transgene expression in plants. Starting from a common set of modules, such as promoters, protein tags and transcribed regions of interest, synthetic genes are assembled, which can be further combined to multigene constructs. As an example, we created T-DNA constructs encoding multiple fluorescent proteins targeted to distinct cellular compartments (nucleus, cytosol, plastids) and demonstrated simultaneous expression of all genes in Nicotiana benthamiana, Lotus japonicus and Arabidopsis thaliana. We assembled an RNA interference (RNAi) module for the construction of intron-spliced hairpin RNA constructs and demonstrated silencing of GFP in N. benthamiana. By combination of the silencing construct together with a codon adapted rescue construct into one vector, our system facilitates genetic complementation and thus confirmation of the causative gene responsible for a given RNAi phenotype. As proof of principle, we silenced a destabilized GFP gene (dGFP) and restored GFP fluorescence by expression of a recoded version of dGFP, which was not targeted by the silencing construct.
Conflict of interest statement
Figures



References
-
- Ahmad P, Ashraf M, Younis M, Hu X, Kumar A, et al. (2012) Role of transgenic plants in agriculture and biopharming. Biotechnology Advances 30: 524–540. - PubMed
-
- Jackson DA, Symons RH, Berg P (1972) Biochemical method for inserting new genetic information into DNA of simian virus 40: Circular SV40 DNA molecules containing lambda phage genes and the galactose operon of Escherichia coli . Proceedings of the National Academy of Sciences, USA 69: 2904–2909. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

