Mycobacterium leprae intracellular survival relies on cholesterol accumulation in infected macrophages: a potential target for new drugs for leprosy treatment
- PMID: 24552180
- PMCID: PMC4262048
- DOI: 10.1111/cmi.12279
Mycobacterium leprae intracellular survival relies on cholesterol accumulation in infected macrophages: a potential target for new drugs for leprosy treatment
Abstract
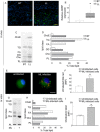
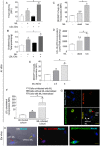
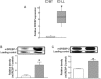
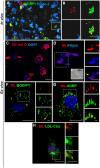
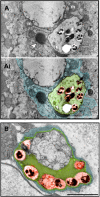
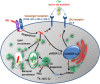
We recently showed that Mycobacterium leprae (ML) is able to induce lipid droplet formation in infected macrophages. We herein confirm that cholesterol (Cho) is one of the host lipid molecules that accumulate in ML-infected macrophages and investigate the effects of ML on cellular Cho metabolism responsible for its accumulation. The expression levels of LDL receptors (LDL-R, CD36, SRA-1, SR-B1, and LRP-1) and enzymes involved in Cho biosynthesis were investigated by qRT-PCR and/or Western blot and shown to be higher in lepromatous leprosy (LL) tissues when compared to borderline tuberculoid (BT) lesions. Moreover, higher levels of the active form of the sterol regulatory element-binding protein (SREBP) transcriptional factors, key regulators of the biosynthesis and uptake of cellular Cho, were found in LL skin biopsies. Functional in vitro assays confirmed the higher capacity of ML-infected macrophages to synthesize Cho and sequester exogenous LDL-Cho. Notably, Cho colocalized to ML-containing phagosomes, and Cho metabolism impairment, through either de novo synthesis inhibition by statins or depletion of exogenous Cho, decreased intracellular bacterial survival. These findings highlight the importance of metabolic integration between the host and bacteria to leprosy pathophysiology, opening new avenues for novel therapeutic strategies to leprosy.
© 2014 The Authors. Cellular Microbiology published by John Wiley & Sons Ltd.
Figures



Similar articles
-
The Essential Role of Cholesterol Metabolism in the Intracellular Survival of Mycobacterium leprae Is Not Coupled to Central Carbon Metabolism and Energy Production.J Bacteriol. 2015 Dec;197(23):3698-707. doi: 10.1128/JB.00625-15. Epub 2015 Sep 21. J Bacteriol. 2015. PMID: 26391209 Free PMC article.
-
Long-term culture of multibacillary leprosy macrophages isolated from skin lesions: a new model to study Mycobacterium leprae-human cell interaction.Br J Dermatol. 2007 Aug;157(2):273-83. doi: 10.1111/j.1365-2133.2007.07992.x. Epub 2007 Jun 6. Br J Dermatol. 2007. PMID: 17553031
-
The function of peroxisome proliferator-activated receptors PPAR-γ and PPAR-δ in Mycobacterium leprae-induced foam cell formation in host macrophages.PLoS Negl Trop Dis. 2020 Oct 19;14(10):e0008850. doi: 10.1371/journal.pntd.0008850. eCollection 2020 Oct. PLoS Negl Trop Dis. 2020. PMID: 33075048 Free PMC article.
-
The immunobiology of leprosy.Int Rev Exp Pathol. 1986;28:45-78. Int Rev Exp Pathol. 1986. PMID: 3516911 Review.
-
New insights into the pathogenesis of leprosy: contribution of subversion of host cell metabolism to bacterial persistence, disease progression, and transmission.F1000Res. 2020 Jan 31;9:F1000 Faculty Rev-70. doi: 10.12688/f1000research.21383.1. eCollection 2020. F1000Res. 2020. PMID: 32051758 Free PMC article. Review.
Cited by
-
Bovine Immunity and Vitamin D3: An Emerging Association in Johne's Disease.Microorganisms. 2022 Sep 18;10(9):1865. doi: 10.3390/microorganisms10091865. Microorganisms. 2022. PMID: 36144467 Free PMC article. Review.
-
Lipid droplets in leukocytes: Organelles linked to inflammatory responses.Exp Cell Res. 2016 Jan 15;340(2):193-7. doi: 10.1016/j.yexcr.2015.10.028. Epub 2015 Oct 29. Exp Cell Res. 2016. PMID: 26515551 Free PMC article. Review.
-
Storage lipid studies in tuberculosis reveal that foam cell biogenesis is disease-specific.PLoS Pathog. 2018 Aug 30;14(8):e1007223. doi: 10.1371/journal.ppat.1007223. eCollection 2018 Aug. PLoS Pathog. 2018. PMID: 30161232 Free PMC article.
-
The formation of lipid droplets favors intracellular Mycobacterium leprae survival in SW-10, non-myelinating Schwann cells.PLoS Negl Trop Dis. 2017 Jun 21;11(6):e0005687. doi: 10.1371/journal.pntd.0005687. eCollection 2017 Jun. PLoS Negl Trop Dis. 2017. PMID: 28636650 Free PMC article.
-
Drug Delivery Systems on Leprosy Therapy: Moving Towards Eradication?Pharmaceutics. 2020 Dec 11;12(12):1202. doi: 10.3390/pharmaceutics12121202. Pharmaceutics. 2020. PMID: 33322356 Free PMC article. Review.
References
-
- Adams LB, Soileau NA, Battista JR. Krahenbuhl JL. Inhibition of metabolism and growth of Mycobacterium leprae by gamma irradiation. Int J Lepr Other Mycobact Dis. 2000;68:1–10. - PubMed
-
- Almeida PE, Silva AR, Maya-Monteiro CM, Töröcsik D, D'Avila H, Dezsö B, et al. Mycobacterium bovis bacillus Calmette-Guérin infection induces TLR2-dependent peroxisome proliferator-activated receptor gamma expression and activation: functions in inflammation, lipid metabolism, and pathogenesis. J Immunol. 2009;183:1337–1345. - PubMed
-
- Almeida PE, Roque NR, Magalhães KG, Mattos KA, Teixeira L, Maya-Monteiro C, et al. Differential TLR2 downstream signaling regulates lipid metabolism and cytokine production triggered by Mycobacterium bovis BCG infection. Biochim Biophys Acta. 2014;1841:97–107. - PubMed
-
- Amaral JJ, Antunes LCM, de Macedo CS, Mattos KA, Han J, Pan J, et al. Metabonomics reveals drastic changes in anti-inflammatory/pro-resolving polyunsaturated fatty acids-derived lipid mediators in leprosy disease. PLoS Negl Trop Dis. 2013;7:e2381. doi: 10.1371/journal.pntd.0002381. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

