Effects of low-dose paroxetine 7.5 mg on weight and sexual function during treatment of vasomotor symptoms associated with menopause
- PMID: 24552977
- PMCID: PMC4175016
- DOI: 10.1097/GME.0000000000000210
Effects of low-dose paroxetine 7.5 mg on weight and sexual function during treatment of vasomotor symptoms associated with menopause
Abstract
Objective: Two phase 3, randomized, placebo-controlled trials demonstrated that low-dose paroxetine 7.5 mg reduced the frequency and severity of vasomotor symptoms (VMS) associated with menopause and had a favorable tolerability profile. The impact of paroxetine 7.5 mg on body weight and sexual function was evaluated in a pooled analysis.
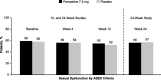
Methods: Postmenopausal women aged 40 years or older who had moderate to severe VMS were randomly assigned to receive paroxetine 7.5 mg or placebo once daily for 12 or 24 weeks. Assessments included changes in body mass index (BMI) and weight, Arizona Sexual Experiences Scale score, Hot Flash-Related Daily Interference Scale sexuality subscore, and adverse events related to weight or sexual dysfunction.
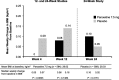
Results: Pooled efficacy and safety populations comprised 1,174 and 1,175 participants, respectively. Baseline values were similar for median weight (∼75 kg), median BMI (∼28 kg/m), and the proportion of women with sexual dysfunction (∼58%). No clinically meaningful or statistically significant changes from baseline in weight or sexual function assessments occurred in the paroxetine 7.5 mg group. Small but statistically significant increases in weight and BMI were observed in the placebo group only on week 4. No significant difference between treatment groups was observed in the proportion of participants who had 7% or higher gain in body weight on week 4, 12, or 24. Rates of adverse events suggestive of sexual dysfunction were low and similar in both treatment groups.
Conclusions: Paroxetine 7.5 mg does not cause weight gain or negative changes in libido when used to treat menopause-associated VMS in postmenopausal women.
Trial registration: ClinicalTrials.gov NCT01101841 NCT01361308.
Figures


Comment in
-
Safety of low-dose paroxetine.Climacteric. 2014 Jun;17(3):313-4. Climacteric. 2014. PMID: 24945035 No abstract available.
-
Another option for menopausal vasomotor symptoms without negative impact on weight and sexual function.Menopause. 2014 Oct;21(10):1036-7. doi: 10.1097/GME.0000000000000309. Menopause. 2014. PMID: 25072955 No abstract available.
References
-
- Williams RE, Kalilani L, DiBenedetti DB, et al. Frequency and severity of vasomotor symptoms among peri- and postmenopausal women in the United States. Climacteric 2008; 11: 32- 43. - PubMed
-
- Ayers B, Hunter MS. Health-related quality of life of women with menopausal hot flushes and night sweats. Climacteric 2013; 16: 235- 239. - PubMed
-
- Williams RE, Kalilani L, DiBenedetti DB, Zhou X, Fehnel SE, Clark RV. Healthcare seeking and treatment for menopausal symptoms in the United States. Maturitas 2007; 58: 348- 358. - PubMed
-
- Hall E, Frey BN, Soares CN. Non-hormonal treatment strategies for vasomotor symptoms: a critical review. Drugs 2011; 71: 287- 304. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

