Extracellular calcium controls the expression of two different forms of ripple-like hippocampal oscillations
- PMID: 24553939
- PMCID: PMC6608517
- DOI: 10.1523/JNEUROSCI.2826-13.2014
Extracellular calcium controls the expression of two different forms of ripple-like hippocampal oscillations
Abstract
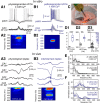
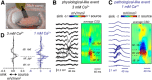
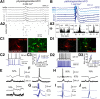
Hippocampal high-frequency oscillations (HFOs) are prominent in physiological and pathological conditions. During physiological ripples (100-200 Hz), few pyramidal cells fire together coordinated by rhythmic inhibitory potentials. In the epileptic hippocampus, fast ripples (>200 Hz) reflect population spikes (PSs) from clusters of bursting cells, but HFOs in the ripple and the fast ripple range are vastly intermixed. What is the meaning of this frequency range? What determines the expression of different HFOs? Here, we used different concentrations of Ca(2+) in a physiological range (1-3 mM) to record local field potentials and single cells in hippocampal slices from normal rats. Surprisingly, we found that this sole manipulation results in the emergence of two forms of HFOs reminiscent of ripples and fast ripples recorded in vivo from normal and epileptic rats, respectively. We scrutinized the cellular correlates and mechanisms underlying the emergence of these two forms of HFOs by combining multisite, single-cell and paired-cell recordings in slices prepared from a rat reporter line that facilitates identification of GABAergic cells. We found a major effect of extracellular Ca(2+) in modulating intrinsic excitability and disynaptic inhibition, two critical factors shaping network dynamics. Moreover, locally modulating the extracellular Ca(2+) concentration in an in vivo environment had a similar effect on disynaptic inhibition, pyramidal cell excitability, and ripple dynamics. Therefore, the HFO frequency band reflects a range of firing dynamics of hippocampal networks.
Keywords: drug delivery; fast ripples; high-frequency oscillations.
Figures






References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
