Boosting functional avidity of CD8+ T cells by vaccinia virus vaccination depends on intrinsic T-cell MyD88 expression but not the inflammatory milieu
- PMID: 24554667
- PMCID: PMC4019089
- DOI: 10.1128/JVI.03664-13
Boosting functional avidity of CD8+ T cells by vaccinia virus vaccination depends on intrinsic T-cell MyD88 expression but not the inflammatory milieu
Abstract
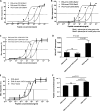
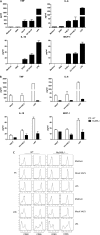
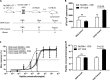
T-cell functional avidity is a crucial determinant for efficient pathogen clearance. Although recombinant DNA priming coupled with a vaccinia-vectored vaccine (VACV) boost has been widely used to mount robust CD8+ T-cell responses, how VACV boost shapes the properties of memory CD8+ T cells remains poorly defined. Here, we characterize the memory CD8+ T cells boosted by VACV and demonstrate that the intrinsic expression of MyD88 is critical for their high functional avidity. Independent of selection of clones with high-affinity T-cell receptor (TCR) or of enhanced proximal TCR signaling, the VACV boost significantly increased T-cell functional avidity through a decrease in the activation threshold. VACV-induced inflammatory milieu is not sufficient for this improvement, as simultaneous administration of the DNA vaccine and mock VACV had no effects on the functional avidity of memory CD8+ T cells. Furthermore, reciprocal adoptive transfer models revealed that the intrinsic MyD88 pathway is required for instructing the functional avidity of CD8+ T cells boosted by VACV. Taking these results together, the intrinsic MyD88 pathway is required for the high functional avidity of VACV-boosted CD8+ T cells independent of TCR selection or the VACV infection-induced MyD88-mediated inflammatory milieu.
Importance: Functional avidity is one of the crucial determinants of T-cell functionality. Interestingly, although it has been demonstrated that a DNA prime-VACV boost regimen elicits high levels of T-cell functional avidity, how VACV changes the low avidity of CD8+ T cells primed by DNA into higher ones in vivo is less defined. Here, we proved that the enhancement of CD8+ T cell avidity induced by VACV boost is mediated by the intrinsic MyD88 pathway but not the MyD88-mediated inflammatory milieu, which might provide prompts in vaccine design.
Figures





Similar articles
-
Immune Signature of Enhanced Functional Avidity CD8+ T Cells in vivo Induced by Vaccinia Vectored Vaccine.Sci Rep. 2017 Feb 3;7:41558. doi: 10.1038/srep41558. Sci Rep. 2017. PMID: 28155878 Free PMC article.
-
The adaptor molecule MyD88 directly promotes CD8 T cell responses to vaccinia virus.J Immunol. 2009 May 15;182(10):6278-86. doi: 10.4049/jimmunol.0803682. J Immunol. 2009. PMID: 19414781 Free PMC article.
-
Langerin+ dermal DC, but not Langerhans cells, are required for effective CD8-mediated immune responses after skin scarification with vaccinia virus.J Invest Dermatol. 2014 Mar;134(3):686-694. doi: 10.1038/jid.2013.418. Epub 2013 Oct 14. J Invest Dermatol. 2014. PMID: 24126845 Free PMC article.
-
Activation and trafficking of CD8+ T cells during viral skin infection: immunological lessons learned from vaccinia virus.Curr Opin Virol. 2018 Feb;28:12-19. doi: 10.1016/j.coviro.2017.10.001. Epub 2017 Oct 25. Curr Opin Virol. 2018. PMID: 29080420 Free PMC article. Review.
-
Uncovering the interplay between CD8, CD4 and antibody responses to complex pathogens.Future Microbiol. 2010 Feb;5(2):221-39. doi: 10.2217/fmb.09.110. Future Microbiol. 2010. PMID: 20143946 Free PMC article. Review.
Cited by
-
The Profile of T Cell Responses in Bacille Calmette-Guérin-Primed Mice Boosted by a Novel Sendai Virus Vectored Anti-Tuberculosis Vaccine.Front Immunol. 2018 Aug 3;9:1796. doi: 10.3389/fimmu.2018.01796. eCollection 2018. Front Immunol. 2018. PMID: 30123219 Free PMC article.
-
Therapeutic Vaccines for the Treatment of HIV.Transl Res. 2020 Sep;223:61-75. doi: 10.1016/j.trsl.2020.04.008. Epub 2020 May 11. Transl Res. 2020. PMID: 32438074 Free PMC article. Review.
-
Prime-Boost Immunization Eliminates Metastatic Colorectal Cancer by Producing High-Avidity Effector CD8+ T Cells.J Immunol. 2017 May 1;198(9):3507-3514. doi: 10.4049/jimmunol.1502672. Epub 2017 Mar 24. J Immunol. 2017. PMID: 28341670 Free PMC article.
-
Intrinsic MyD88-Akt1-mTOR Signaling Coordinates Disparate Tc17 and Tc1 Responses during Vaccine Immunity against Fungal Pneumonia.PLoS Pathog. 2015 Sep 14;11(9):e1005161. doi: 10.1371/journal.ppat.1005161. eCollection 2015 Sep. PLoS Pathog. 2015. PMID: 26367276 Free PMC article.
-
Cancer testis antigen Cyclin A1 harbors several HLA-A*02:01-restricted T cell epitopes, which are presented and recognized in vivo.Cancer Immunol Immunother. 2020 Jul;69(7):1217-1227. doi: 10.1007/s00262-020-02519-6. Epub 2020 Mar 10. Cancer Immunol Immunother. 2020. PMID: 32157447 Free PMC article.
References
-
- Harari A, Bart PA, Stohr W, Tapia G, Garcia M, Medjitna-Rais E, Burnet S, Cellerai C, Erlwein O, Barber T, Moog C, Liljestrom P, Wagner R, Wolf H, Kraehenbuhl JP, Esteban M, Heeney J, Frachette MJ, Tartaglia J, McCormack S, Babiker A, Weber J, Pantaleo G. 2008. An HIV-1 clade C DNA prime, NYVAC boost vaccine regimen induces reliable, polyfunctional, and long-lasting T cell responses. J. Exp. Med. 205:63–77. 10.1084/jem.20071331 - DOI - PMC - PubMed
-
- Sandstrom E, Nilsson C, Hejdeman B, Brave A, Bratt G, Robb M, Cox J, Vancott T, Marovich M, Stout R, Aboud S, Bakari M, Pallangyo K, Ljungberg K, Moss B, Earl P, Michael N, Birx D, Mhalu F, Wahren B, Biberfeld G. 2008. Broad immunogenicity of a multigene, multiclade HIV-1 DNA vaccine boosted with heterologous HIV-1 recombinant modified vaccinia virus Ankara. J. Infect. Dis. 198:1482–1490. 10.1086/592507 - DOI - PMC - PubMed
-
- Kibuuka H, Kimutai R, Maboko L, Sawe F, Schunk MS, Kroidl A, Shaffer D, Eller LA, Kibaya R, Eller MA, Schindler KB, Schuetz A, Millard M, Kroll J, Dally L, Hoelscher M, Bailer R, Cox JH, Marovich M, Birx DL, Graham BS, Michael NL, de Souza MS, Robb ML. 2010. A phase 1/2 study of a multiclade HIV-1 DNA plasmid prime and recombinant adenovirus serotype 5 boost vaccine in HIV-uninfected east Africans (RV 172). J. Infect. Dis. 201:600–607. 10.1086/650299 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

