Systemic stress signalling: understanding the cell non-autonomous control of proteostasis
- PMID: 24556842
- PMCID: PMC5922984
- DOI: 10.1038/nrm3752
Systemic stress signalling: understanding the cell non-autonomous control of proteostasis
Abstract
Proteome maintenance is crucial to cellular health and viability, and is typically thought to be controlled in a cell-autonomous manner. However, recent evidence indicates that protein-folding defects can systemically activate proteostasis mechanisms through signalling pathways that coordinate stress responses among tissues. Coordination of ageing rates between tissues may also be mediated by systemic modulation of proteostasis. These findings suggest that proteome maintenance is a systemically regulated process, a discovery that may have important therapeutic implications.
Conflict of interest statement
The authors declare no competing interests.
Figures



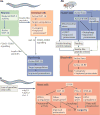

References
-
- Balch WE, Morimoto RI, Dillin A, Kelly JW. Adapting proteostasis for disease intervention. Science. 2008;319:916–919. - PubMed
-
- Poon HF, Vaishnav RA, Getchell TV, Getchell ML, Butterfield DA. Quantitative proteomics analysis of differential protein expression and oxidative modification of specific proteins in the brains of old mice. Neurobiol. Aging. 2006;27:1010–1019. - PubMed
-
- Cotto JJ, Morimoto RI. Stress-induced activation of the heat-shock response: cell and molecular biology of heat-shock factors. Biochem. Soc. Symp. 1999;64:105–118. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

