Cryptococcus neoformans glucuronoxylomannan fractions of different molecular masses are functionally distinct
- PMID: 24571070
- PMCID: PMC4294706
- DOI: 10.2217/fmb.13.163
Cryptococcus neoformans glucuronoxylomannan fractions of different molecular masses are functionally distinct
Abstract
Aims: Glucuronoxylomannan (GXM) is the major polysaccharide component of Cryptococcus neoformans. We evaluated in this study whether GXM fractions of different molecular masses were functionally distinct.
Materials & methods: GXM samples isolated from C. neoformans cultures were fractionated to generate polysaccharide preparations differing in molecular mass. These fractions were used in experiments focused on the association of GXM with cell wall components of C. neoformans, as well as on the interaction of the polysaccharide with host cells.
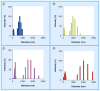
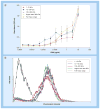
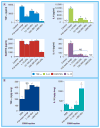
Results & conclusion: GXM fractions of variable molecular masses bound to the surface of a C. neoformans acapsular mutant in a punctate pattern that is in contrast to the usual annular pattern of surface coating observed when GXM samples containing the full molecular mass range were used. The polysaccharide samples were also significantly different in their ability to stimulate cytokine production by host cells. Our findings indicate that GXM fractions are functionally distinct depending on their mass.
Figures



Similar articles
-
Binding of the wheat germ lectin to Cryptococcus neoformans chitooligomers affects multiple mechanisms required for fungal pathogenesis.Fungal Genet Biol. 2013 Nov;60:64-73. doi: 10.1016/j.fgb.2013.04.005. Epub 2013 Apr 19. Fungal Genet Biol. 2013. PMID: 23608320 Free PMC article.
-
A Paracoccidioides brasiliensis glycan shares serologic and functional properties with cryptococcal glucuronoxylomannan.Fungal Genet Biol. 2012 Nov;49(11):943-54. doi: 10.1016/j.fgb.2012.09.002. Epub 2012 Sep 23. Fungal Genet Biol. 2012. PMID: 23010152 Free PMC article.
-
Evasion of Innate Immune Responses by the Highly Virulent Cryptococcus gattii by Altering Capsule Glucuronoxylomannan Structure.Front Cell Infect Microbiol. 2016 Jan 6;5:101. doi: 10.3389/fcimb.2015.00101. eCollection 2015. Front Cell Infect Microbiol. 2016. PMID: 26779451 Free PMC article.
-
The capsule of the fungal pathogen Cryptococcus neoformans.Adv Appl Microbiol. 2009;68:133-216. doi: 10.1016/S0065-2164(09)01204-0. Adv Appl Microbiol. 2009. PMID: 19426855 Free PMC article. Review.
-
Polysaccharides, mimotopes and vaccines for fungal and encapsulated pathogens.Trends Microbiol. 2001 Sep;9(9):445-51. doi: 10.1016/s0966-842x(01)02134-5. Trends Microbiol. 2001. PMID: 11553457 Review.
Cited by
-
The mechanical properties of microbial surfaces and biofilms.Cell Surf. 2019 Aug 5;5:100028. doi: 10.1016/j.tcsw.2019.100028. eCollection 2019 Dec. Cell Surf. 2019. PMID: 32743144 Free PMC article.
-
Dexamethasone and Methylprednisolone Promote Cell Proliferation, Capsule Enlargement, and in vivo Dissemination of C. neoformans.Front Fungal Biol. 2021 Feb 10;2:643537. doi: 10.3389/ffunb.2021.643537. eCollection 2021. Front Fungal Biol. 2021. PMID: 37744119 Free PMC article.
-
Neutron Scattering Analysis of Cryptococcus neoformans Polysaccharide Reveals Solution Rigidity and Repeating Fractal-like Structural Patterns.Biomacromolecules. 2024 Feb 12;25(2):690-699. doi: 10.1021/acs.biomac.3c00911. Epub 2023 Dec 29. Biomacromolecules. 2024. PMID: 38157431 Free PMC article.
-
Non-self glycan structures as possible modulators of cancer progression: would polysaccharides from Cryptococcus spp. impact this phenomenon?Braz J Microbiol. 2023 Jun;54(2):907-919. doi: 10.1007/s42770-023-00936-0. Epub 2023 Feb 25. Braz J Microbiol. 2023. PMID: 36840821 Free PMC article. Review.
-
Virulence of Cryptococcus sp. Biofilms In Vitro and In Vivo using Galleria mellonella as an Alternative Model.Front Microbiol. 2016 Mar 9;7:290. doi: 10.3389/fmicb.2016.00290. eCollection 2016. Front Microbiol. 2016. PMID: 27014214 Free PMC article.
References
-
- Taylor CM, Roberts IS. Capsular polysaccharides and their role in virulence. Contrib Microbiol. 2005;12:55–66. - PubMed
-
- Guo H, Yi W, Song JK, Wang PG. Current understanding on biosynthesis of microbial polysaccharides. Curr Top Med Chem. 2008;8(2):141–151. - PubMed
-
- Da Silva CA, Chalouni C, Williams A, Hartl D, Lee CG, Elias JA. Chitin is a size-dependent regulator of macrophage TNF and IL-10 production. J Immunol. 2009;182(6):3573–3582. First evidence in the literature showing that polysaccharide dimensions correlated with immunological activity. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
