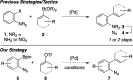
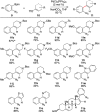
Development of a Suzuki cross-coupling reaction between 2-azidoarylboronic pinacolate esters and vinyl triflates to enable the synthesis of [2,3]-fused indole heterocycles
- PMID: 24571492
- PMCID: PMC3985933
- DOI: 10.1021/jo500252e
Development of a Suzuki cross-coupling reaction between 2-azidoarylboronic pinacolate esters and vinyl triflates to enable the synthesis of [2,3]-fused indole heterocycles
Abstract
The scope and limitations of a Suzuki reaction between 2-azidoarylboronic acid pinacolate esters and vinyl triflates are reported. This cross-coupling reaction enables the regioselective synthesis of indoles after a subsequent Rh(II)2-catalyzed sp(2)-C-H bond amination reaction.
Figures
Similar articles
-
A palladium-catalyzed three-component cross-coupling of conjugated dienes or terminal alkenes with vinyl triflates and boronic acids.J Am Chem Soc. 2011 Apr 20;133(15):5784-7. doi: 10.1021/ja201358b. Epub 2011 Mar 30. J Am Chem Soc. 2011. PMID: 21449562 Free PMC article.
-
Palladium-Catalyzed Methylation of Aryl, Heteroaryl, and Vinyl Boronate Esters.Org Lett. 2019 Mar 1;21(5):1337-1341. doi: 10.1021/acs.orglett.9b00025. Epub 2019 Feb 14. Org Lett. 2019. PMID: 30763109 Free PMC article.
-
Tandem intermolecular Suzuki coupling/intramolecular vinyl triflate-arene coupling.Chem Commun (Camb). 2002 Apr 21;(8):832-3. doi: 10.1039/b200692h. Chem Commun (Camb). 2002. PMID: 12123001
-
[Synthesis of indole compounds using multifunctional synthons].Yakugaku Zasshi. 2013;133(1):99-106. doi: 10.1248/yakushi.12-00233. Yakugaku Zasshi. 2013. PMID: 23292026 Review. Japanese.
-
Continuous Flow Synthesis of Heterocycles: A Recent Update on the Flow Synthesis of Indoles.Molecules. 2020 Jul 16;25(14):3242. doi: 10.3390/molecules25143242. Molecules. 2020. PMID: 32708643 Free PMC article. Review.
Cited by
-
Radical Functionalization of Unsaturated Amino Acids: Synthesis of Side-Chain-Fluorinated, Azido-Substituted, and Hydroxylated Amino Acids.ACS Omega. 2019 Jun 21;4(6):10854-10865. doi: 10.1021/acsomega.9b01509. eCollection 2019 Jun 30. ACS Omega. 2019. PMID: 31460183 Free PMC article.
-
Time-Gated Detection of Cystathionine γ-Lyase Activity and Inhibition with a Selective, Luminogenic Hydrogen Sulfide Sensor.Chemistry. 2017 Jan 18;23(4):752-756. doi: 10.1002/chem.201604786. Epub 2016 Nov 25. Chemistry. 2017. PMID: 27734530 Free PMC article.
-
Insights Into the Detection Selectivity of Redox and Non-redox Based Probes for the Superoxide Anion Using Coumarin and Chromone as the Fluorophores.Front Chem. 2021 Nov 25;9:753621. doi: 10.3389/fchem.2021.753621. eCollection 2021. Front Chem. 2021. PMID: 34912779 Free PMC article.
References
-
-
For reviews, see:
- Gribble G. W.; Saulnier M. G.; Pelkey E. T.; Kishbaugh T. L. S.; Liu Y.; Jiang J.; Trujillo H. A.; Keavy D. J.; Davis D. A.; Conway S. C.; Switzer F. L.; Roy S.; Silva R. A.; Obaza-Nutaitis J. A.; Sibi M. P.; Moskalev N. V.; Barden T. C.; Chang L.; Habeski (née Simon) W. M.; Pelcman B.; Sponholtz W. R. III; Chau R. W.; Allison B. D.; Garaas S. D.; Sinha M. S.; McGowan M. A.; Reese M. R.; Harpp K. S. Curr. Org. Chem. 2005, 9, 1493.
- Agarwal S.; Cämmerer S.; Filali S.; Fröhner W.; Knöll J.; Krahl M. P.; Reddy K. R.; Knölker H.-J. Curr. Org. Chem. 2005, 9, 1601.
- Crich D.; Banerjee A. Acc. Chem. Res. 2007, 40, 151. - PubMed
- Edwankar C. R.; Edwankar R. V.; Namjoshi O. A.; Rallapappi S. K.; Yang J.; Cook J. M. Curr. Opin. Drug Discuss. Dev. 2009, 12, 752. - PubMed
- Bandini M.; Eichholzer A. Angew. Chem., Int. Ed. 2009, 48, 9608. - PubMed
- Ruiz-Sanchis P.; Savina S. A.; Albericio F.; Álvarez M. Chem. Eur. J. 2011, 17, 1388. - PubMed
- Taber D. F.; Tirunahari P. K. Tetrahedron 2011, 67, 7195. - PMC - PubMed
- Schmidt A. W.; Reddy K. R.; Knölker H.-J. Chem. Rev. 2012, 112, 3193. - PubMed
-
-
-
For examples of their occurrence in electronic organic materials, see:
- van Dijken A.; Bastiaansen J. J. A. M.; Kiggen N. M. M.; Langeveld B. M. W.; Rothe C.; Monkman A.; Bach I.; Stössel P.; Brunner K. J. Am. Chem. Soc. 2004, 126, 7718. - PubMed
- Wu Y.; Li Y.; Gardner S.; Ong B. S. J. Am. Chem. Soc. 2005, 127, 614. - PubMed
- Boudreault P.-L. T.; Wakim S.; Blouin N.; Simard M.; Tessier C.; Tao Y.; Leclerc M. J. Am. Chem. Soc. 2007, 129, 9125. - PubMed
- Wang C.; Dong H.; Hu W.; Liu Y.; Zhu D. Chem. Rev. 2011, 112, 2208. - PubMed
-
-
- Stokes B. J.; Dong H.; Leslie B. E.; Pumphrey A. L.; Driver T. G. J. Am. Chem. Soc. 2007, 129, 7500. - PubMed
- Shen M.; Leslie B. E.; Driver T. G. Angew. Chem., Int. Ed. 2008, 47, 5056. - PubMed
- Stokes B. J.; Jovanović B.; Dong H.; Richert K. J.; Riell R. D.; Driver T. G. J. Org. Chem. 2009, 74, 3225. - PMC - PubMed
- Pumphrey A. L.; Dong H.; Driver T. G. Angew. Chem., Int. Ed. 2012, 51, 5920. - PMC - PubMed
-
-
For reviews on the preparation of vinyl or aryl azides, see:
- L’abbé G. Angew. Chem., Int. Ed. 1975, 14, 775.
- Scriven E. F. V.; Turnbull K. Chem. Rev. 1988, 88, 297.
- Bräse S.; Gil C.; Knepper K.; Zimmermann V. Angew. Chem., Int. Ed. 2005, 44, 5188. - PubMed
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources