Monoamine oxidase is a major determinant of redox balance in human atrial myocardium and is associated with postoperative atrial fibrillation
- PMID: 24572256
- PMCID: PMC3959694
- DOI: 10.1161/JAHA.113.000713
Monoamine oxidase is a major determinant of redox balance in human atrial myocardium and is associated with postoperative atrial fibrillation
Abstract
Background: Onset of postoperative atrial fibrillation (POAF) is a common and costly complication of heart surgery despite major improvements in surgical technique and quality of patient care. The etiology of POAF, and the ability of clinicians to identify and therapeutically target high-risk patients, remains elusive.
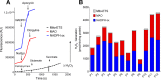
Methods and results: Myocardial tissue dissected from right atrial appendage (RAA) was obtained from 244 patients undergoing cardiac surgery. Reactive oxygen species (ROS) generation from multiple sources was assessed in this tissue, along with total glutathione (GSHt) and its related enzymes GSH-peroxidase (GPx) and GSH-reductase (GR). Monoamine oxidase (MAO) and NADPH oxidase were observed to generate ROS at rates 10-fold greater than intact, coupled mitochondria. POAF risk was significantly associated with MAO activity (Quartile 1 [Q1]: adjusted relative risk [ARR]=1.0; Q2: ARR=1.8, 95% confidence interval [CI]=0.84 to 4.0; Q3: ARR=2.1, 95% CI=0.99 to 4.3; Q4: ARR=3.8, 95% CI=1.9 to 7.5; adjusted Ptrend=0.009). In contrast, myocardial GSHt was inversely associated with POAF (Quartile 1 [Q1]: adjusted relative risk [ARR]=1.0; Q2: ARR=0.93, 95% confidence interval [CI]=0.60 to 1.4; Q3: ARR=0.62, 95% CI=0.36 to 1.1; Q4: ARR=0.56, 95% CI=0.34 to 0.93; adjusted Ptrend=0.014). GPx also was significantly associated with POAF; however, a linear trend for risk was not observed across increasing levels of the enzyme. GR was not associated with POAF risk.
Conclusions: Our results show that MAO is an important determinant of redox balance in human atrial myocardium, and that this enzyme, in addition to GSHt and GPx, is associated with an increased risk for POAF. Further investigation is needed to validate MAO as a predictive biomarker for POAF, and to explore this enzyme's potential role in arrhythmogenesis.
Keywords: biomarkers; cardiopulmonary bypass; catecholamines; oxidative stress; post‐operative atrial fibrillation; redox; tachyarrhythmias.
Figures



References
-
- Mathew JP, Fontes ML, Tudor IC, Ramsay J, Duke P, Mazer CD, Barash PG, Hsu PH, Mangano DT. A multicenter risk index for atrial fibrillation after cardiac surgery. JAMA. 2004; 291:1720-1729 - PubMed
-
- Ahlsson A, Fengsrud E, Bodin L, Englund A. Postoperative atrial fibrillation in patients undergoing aortocoronary bypass surgery carries an eightfold risk of future atrial fibrillation and a doubled cardiovascular mortality. Eur J Cardiothorac Surg. 2010; 37:1353-1359 - PubMed
-
- El‐Chami MF, Kilgo P, Thourani V, Lattouf OM, Delurgio DB, Guyton RA, Leon AR, Puskas JD. New‐onset atrial fibrillation predicts long‐term mortality after coronary artery bypass graft. J Am Coll Cardiol. 2010; 55:1370-1376 - PubMed
-
- Aviles RJ, Martin DO, Apperson‐Hansen C, Houghtaling PL, Rautaharju P, Kronmal RA, Tracy RP, Van Wagoner DR, Psaty BM, Lauer MS, Chung MK. Inflammation as a risk factor for atrial fibrillation. Circulation. 2003; 108:3006-3010 - PubMed
-
- Ishii Y, Schuessler RB, Gaynor SL, Yamada K, Fu AS, Boineau JP, Damiano RJ., Jr Inflammation of atrium after cardiac surgery is associated with inhomogeneity of atrial conduction and atrial fibrillation. Circulation. 2005; 111:2881-2888 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

