Absence of transient receptor potential vanilloid-1 accelerates stress-induced axonopathy in the optic projection
- PMID: 24573275
- PMCID: PMC3935081
- DOI: 10.1523/JNEUROSCI.4089-13.2014
Absence of transient receptor potential vanilloid-1 accelerates stress-induced axonopathy in the optic projection
Abstract
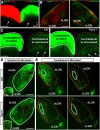
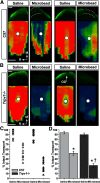
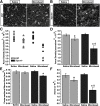
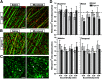
How neurons respond to stress in degenerative disease is of fundamental importance for identifying mechanisms of progression and new therapeutic targets. Members of the transient receptor potential (TRP) family of cation-selective ion channels are candidates for mediating stress signals, since different subunits transduce a variety of stimuli relevant in both normal and pathogenic physiology. We addressed this possibility for the TRP vanilloid-1 (TRPV1) subunit by comparing how the optic projection of Trpv1(-/-) mice and age-matched C57 controls responds to stress from elevated ocular pressure, the critical stressor in the most common optic neuropathy, glaucoma. Over a 5 week period of elevated pressure induced by microbead occlusion of ocular fluid, Trpv1(-/-) accelerated both degradation of axonal transport from retinal ganglion cells to the superior colliculus and degeneration of the axons themselves in the optic nerve. Ganglion cell body loss, which is normally later in progression, occurred in nasal sectors of Trpv1(-/-) but not C57 retina. Pharmacological antagonism of TRPV1 in rats similarly accelerated ganglion cell axonopathy. Elevated ocular pressure resulted in differences in spontaneous firing rate and action potential threshold current in Trpv1(-/-) ganglion cells compared with C57. In the absence of elevated pressure, ganglion cells in the two strains had similar firing patterns. Based on these data, we propose that TRPV1 may help neurons respond to disease-relevant stressors by enhancing activity necessary for axonal signaling.
Keywords: TRPV1; glaucoma; microbead glaucoma; optic nerve; retinal ganglion cell; superior colliculus.
Figures

Similar articles
-
Short-term increases in transient receptor potential vanilloid-1 mediate stress-induced enhancement of neuronal excitation.J Neurosci. 2014 Nov 12;34(46):15369-81. doi: 10.1523/JNEUROSCI.3424-14.2014. J Neurosci. 2014. PMID: 25392504 Free PMC article.
-
Activation of transient receptor potential vanilloid-1 (TRPV1) influences how retinal ganglion cell neurons respond to pressure-related stress.Channels (Austin). 2015;9(2):102-13. doi: 10.1080/19336950.2015.1009272. Channels (Austin). 2015. PMID: 25713995 Free PMC article.
-
Proximal inhibition of p38 MAPK stress signaling prevents distal axonopathy.Neurobiol Dis. 2013 Nov;59:26-37. doi: 10.1016/j.nbd.2013.07.001. Epub 2013 Jul 13. Neurobiol Dis. 2013. PMID: 23859799 Free PMC article.
-
Critical pathogenic events underlying progression of neurodegeneration in glaucoma.Prog Retin Eye Res. 2012 Nov;31(6):702-19. doi: 10.1016/j.preteyeres.2012.07.001. Epub 2012 Aug 1. Prog Retin Eye Res. 2012. PMID: 22871543 Free PMC article. Review.
-
Assessment of retinal ganglion cell damage in glaucomatous optic neuropathy: Axon transport, injury and soma loss.Exp Eye Res. 2015 Dec;141:111-24. doi: 10.1016/j.exer.2015.06.006. Epub 2015 Jun 9. Exp Eye Res. 2015. PMID: 26070986 Review.
Cited by
-
Involvement of transient receptor potential channels in ocular diseases: a narrative review.Ann Transl Med. 2022 Aug;10(15):839. doi: 10.21037/atm-21-6145. Ann Transl Med. 2022. PMID: 36034986 Free PMC article.
-
Effects of ocular hypertension in the visual system of pigmented mice.PLoS One. 2015 Mar 26;10(3):e0121134. doi: 10.1371/journal.pone.0121134. eCollection 2015. PLoS One. 2015. PMID: 25811653 Free PMC article.
-
Towards A Microbead Occlusion Model of Glaucoma for a Non-Human Primate.Sci Rep. 2019 Aug 9;9(1):11572. doi: 10.1038/s41598-019-48054-y. Sci Rep. 2019. PMID: 31399621 Free PMC article.
-
Progression and Pathology of Traumatic Optic Neuropathy From Repeated Primary Blast Exposure.Front Neurosci. 2019 Jul 11;13:719. doi: 10.3389/fnins.2019.00719. eCollection 2019. Front Neurosci. 2019. PMID: 31354422 Free PMC article.
-
Oral Delivery of a Synthetic Sterol Reduces Axonopathy and Inflammation in a Rodent Model of Glaucoma.Front Neurosci. 2017 Feb 7;11:45. doi: 10.3389/fnins.2017.00045. eCollection 2017. Front Neurosci. 2017. PMID: 28223915 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
