Antibodies that detect O-linked β-D-N-acetylglucosamine on the extracellular domain of cell surface glycoproteins
- PMID: 24573683
- PMCID: PMC4036252
- DOI: 10.1074/jbc.M113.492512
Antibodies that detect O-linked β-D-N-acetylglucosamine on the extracellular domain of cell surface glycoproteins
Abstract
The transfer of N-acetylglucosamine (GlcNAc) to Ser or Thr in cytoplasmic and nuclear proteins is a well known post-translational modification that is catalyzed by the O-GlcNAc transferase OGT. A more recently identified O-GlcNAc transferase, EOGT, functions in the secretory pathway and transfers O-GlcNAc to proteins with epidermal growth factor-like (EGF) repeats. A number of antibodies that detect O-GlcNAc in cytosolic and nuclear extracts have been described previously. Here we compare seven of these antibodies (CTD110.6, 10D8, RL2, HGAC85, 18B10.C7(#3), 9D1.E4(#10), and 1F5.D6 (#14) for detection of the O-GlcNAc modification on extracellular domains of membrane or secreted glycoproteins that may also carry various N- and O-glycans. We found that CTD110.6 binds not only to O-GlcNAc on proteins but also to terminal β-GlcNAc on the complex N-glycans of Lec8 Chinese hamster ovary (CHO) cells that lack UDP-Gal transporter activity and express GlcNAc-terminating, complex N-glycans. We show that CTD110.6, #3, and #10 antibodies can be used to detect cell surface glycoproteins bearing O-GlcNAc. Cell surface glycoproteins recognized by CTD110.6 antibody included NOTCH1 that possesses many EGF repeats with a consensus site for EOGT. Knockdown of CHO Eogt reduced binding of CTD110.6 to Lec1 CHO cells, and expression of a human EOGT cDNA increased the O-GlcNAc signal on Lec1 cells and the extracellular domain of NOTCH1. Thus, with careful controls, antibodies CTD110.6 (IgM), #3 (IgG), and #10 (IgG) can be used to detect membrane and secreted proteins modified by O-GlcNAc on EGF repeats.
Keywords: Antibodies; CHO; Flow Cytometry; GlcNAc-terminating N-Glycans; Lec1; Lec8; Mutant; O-GlcNAc; O-GlcNAcylation.
Figures


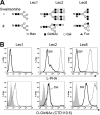
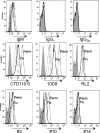

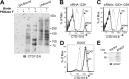

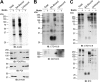
References
-
- Varki A., Cummings R. D., Esko J. D., Freeze H. H., Stanley P., Bertozzi C. R., Hart G. W., Etlzler M. E. (2009) Essentials of Glycobiology, 2nd Edition, Cold Spring Harbor Laboratory, Cold Spring Harbor, NY - PubMed
-
- Matsuura A., Ito M., Sakaidani Y., Kondo T., Murakami K., Furukawa K., Nadano D., Matsuda T., Okajima T. (2008) O-Linked N-acetylglucosamine is present on the extracellular domain of Notch receptors. J. Biol. Chem. 283, 35486–35495 - PubMed
-
- Sakaidani Y., Furukawa K., Okajima T. (2010) O-GlcNAc modification of the extracellular domain of Notch receptors. Methods Enzymol. 480, 355–373 - PubMed
-
- Sakaidani Y., Ichiyanagi N., Saito C., Nomura T., Ito M., Nishio Y., Nadano D., Matsuda T., Furukawa K., Okajima T. (2012) O-Linked N-acetylglucosamine modification of mammalian Notch receptors by an atypical O-GlcNAc transferase Eogt1. Biochem. Biophys. Res. Commun. 419, 14–19 - PubMed
-
- Sakaidani Y., Nomura T., Matsuura A., Ito M., Suzuki E., Murakami K., Nadano D., Matsuda T., Furukawa K., Okajima T. (2011) O-Linked-N-acetylglucosamine on extracellular protein domains mediates epithelial cell-matrix interactions. Nat. Commun. 2, 583. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

