Cancer stem cells in colorectal cancer from pathogenesis to therapy: controversies and perspectives
- PMID: 24574766
- PMCID: PMC3921545
- DOI: 10.3748/wjg.v20.i4.923
Cancer stem cells in colorectal cancer from pathogenesis to therapy: controversies and perspectives
Abstract
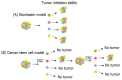
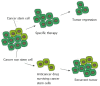
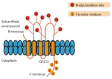
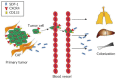
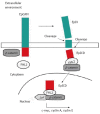
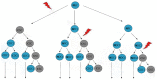
Colorectal cancer remains one of the most common and lethal malignancies worldwide despite the use of various therapeutic strategies. A better understanding of the mechanisms responsible for tumor initiation and progression is essential for the development of novel, more powerful therapies. The traditional, so-called "stochastic model" of tumor development, which assumes that each cancer cell is tumorigenic, has been deeply challenged during the past decade by the identification of cancer stem cells (CSCs), a biologically distinct subset of cells within the bulk of tumor mass. This discovery led to the development of the hierarchical model of tumorigenesis which assumes that only CSCs have the ability to initiate tumor growth, both at primary and metastatic sites. This model implies that the elimination of all CSCs is fundamental to eradicate tumors and that failure to do so might be responsible for the occurrence of relapses and/or metastases frequently observed in the clinical management of colorectal cancer patients. Identification and isolation of CSCs is essential for a better understanding of their role in the tumorigenetic process and for the development of CSC-specific therapies. Several methods have been used for this purpose and many efforts have been focused on the identification of specific CSC-surface markers. This review provides an overview of the proposed roles of CSC in human colorectal tumorigenesis focusing on the most important molecules identified as CSC-specific markers in colorectal cancer and on the potential strategies for the development of CSC-targeted therapy.
Keywords: Cancer stem cells; Cancer therapy; Colorectal cancer; Prognostic marker; Tumorigenesis.
Figures


Similar articles
-
Targeting cancer stem-like cells in glioblastoma and colorectal cancer through metabolic pathways.Int J Cancer. 2017 Jan 1;140(1):10-22. doi: 10.1002/ijc.30259. Epub 2016 Jul 20. Int J Cancer. 2017. PMID: 27389307 Review.
-
Prostaglandin E2 Promotes Colorectal Cancer Stem Cell Expansion and Metastasis in Mice.Gastroenterology. 2015 Dec;149(7):1884-1895.e4. doi: 10.1053/j.gastro.2015.07.064. Epub 2015 Aug 7. Gastroenterology. 2015. PMID: 26261008 Free PMC article.
-
Characterization of cancer stem cells from different grades of human colorectal cancer.Tumour Biol. 2016 Oct;37(10):14069-14081. doi: 10.1007/s13277-016-5232-6. Epub 2016 Aug 9. Tumour Biol. 2016. PMID: 27507615
-
Cancer Stem Cell-Based Models of Colorectal Cancer Reveal Molecular Determinants of Therapy Resistance.Stem Cells Transl Med. 2016 Apr;5(4):511-23. doi: 10.5966/sctm.2015-0214. Epub 2016 Mar 8. Stem Cells Transl Med. 2016. PMID: 26956206 Free PMC article.
-
Colon cancer stem cells: controversies and perspectives.World J Gastroenterol. 2013 May 28;19(20):2997-3006. doi: 10.3748/wjg.v19.i20.2997. World J Gastroenterol. 2013. PMID: 23716979 Free PMC article. Review.
Cited by
-
Haploinsufficiency of the c-myc transcriptional repressor FIR, as a dominant negative-alternative splicing model, promoted p53-dependent T-cell acute lymphoblastic leukemia progression by activating Notch1.Oncotarget. 2015 Mar 10;6(7):5102-17. doi: 10.18632/oncotarget.3244. Oncotarget. 2015. PMID: 25671302 Free PMC article.
-
Biomarkers and signaling pathways of colorectal cancer stem cells.Tumour Biol. 2015 Mar;36(3):1339-53. doi: 10.1007/s13277-015-3198-4. Epub 2015 Feb 14. Tumour Biol. 2015. PMID: 25680406 Review.
-
Cancer stem cell theory and the warburg effect, two sides of the same coin?Int J Mol Sci. 2014 May 19;15(5):8893-930. doi: 10.3390/ijms15058893. Int J Mol Sci. 2014. PMID: 24857919 Free PMC article.
-
LncRNA LINC01315 silencing modulates cancer stem cell properties and epithelial-to-mesenchymal transition in colorectal cancer via miR-484/DLK1 axis.Cell Cycle. 2022 Apr;21(8):851-873. doi: 10.1080/15384101.2022.2033415. Epub 2022 Feb 13. Cell Cycle. 2022. PMID: 35156543 Free PMC article.
-
Knockdown of a DIS3L2 promoter upstream long noncoding RNA (AC105461.1) enhances colorectal cancer stem cell properties in vitro by down-regulating DIS3L2.Onco Targets Ther. 2017 May 2;10:2367-2376. doi: 10.2147/OTT.S132708. eCollection 2017. Onco Targets Ther. 2017. PMID: 28496335 Free PMC article.
References
-
- Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JW, Comber H, Forman D, Bray F. Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer. 2013;49:1374–1403. - PubMed
-
- McDonald SA, Preston SL, Lovell MJ, Wright NA, Jankowski JA. Mechanisms of disease: from stem cells to colorectal cancer. Nat Clin Pract Gastroenterol Hepatol. 2006;3:267–274. - PubMed
-
- Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature. 2007;449:1003–1007. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

