Diagnostic approach to the congenital muscular dystrophies
- PMID: 24581957
- PMCID: PMC5258110
- DOI: 10.1016/j.nmd.2013.12.011
Diagnostic approach to the congenital muscular dystrophies
Abstract
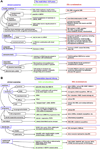
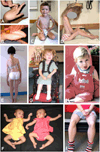
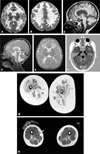
Congenital muscular dystrophies (CMDs) are early onset disorders of muscle with histological features suggesting a dystrophic process. The congenital muscular dystrophies as a group encompass great clinical and genetic heterogeneity so that achieving an accurate genetic diagnosis has become increasingly challenging, even in the age of next generation sequencing. In this document we review the diagnostic features, differential diagnostic considerations and available diagnostic tools for the various CMD subtypes and provide a systematic guide to the use of these resources for achieving an accurate molecular diagnosis. An International Committee on the Standard of Care for Congenital Muscular Dystrophies composed of experts on various aspects relevant to the CMDs performed a review of the available literature as well as of the unpublished expertise represented by the members of the committee and their contacts. This process was refined by two rounds of online surveys and followed by a three-day meeting at which the conclusions were presented and further refined. The combined consensus summarized in this document allows the physician to recognize the presence of a CMD in a child with weakness based on history, clinical examination, muscle biopsy results, and imaging. It will be helpful in suspecting a specific CMD subtype in order to prioritize testing to arrive at a final genetic diagnosis.
Keywords: Alpha-dystroglycan; Collagen VI; Congenital muscular dystrophy; Diagnostic guideline; Lamin A/C; Laminin alpha2; RYR1; SEPN1.
Copyright © 2014 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
Authors declared that there is no conflict of interest.
Figures

References
-
- Nonaka I, Une Y, Ishihara T, Miyoshino S, Nakashima T, Sugita H. A clinical and histological study of Ullrich’s disease (congenital atonic-sclerotic muscular dystrophy) Neuropediatrics. 1981;12:197–208. - PubMed
-
- Kobayashi O, Hayashi Y, Arahata K, Ozawa E, Nonaka I. Congenital muscular dystrophy: clinical and pathologic study of 50 patients with the classical (Occidental) merosin-positive form. Neurology. 1996;46:815–818. - PubMed
-
- Reed UC. Congenital muscular dystrophy. Part I: a review of phenotypical and diagnostic aspects. Arq Neuropsiquiatr. 2009;67:144–168. - PubMed
-
- Reed UC. Congenital muscular dystrophy. Part II: a review of pathogenesis and therapeutic perspectives. Arq Neuropsiquiatr. 2009;67:343–362. - PubMed
-
- Mercuri E, Muntoni F. The ever-expanding spectrum of congenital muscular dystrophies. Ann Neurol. 2012;72:9–17. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

