Membrane potential-dependent uptake of 18F-triphenylphosphonium--a new voltage sensor as an imaging agent for detecting burn-induced apoptosis
- PMID: 24582214
- PMCID: PMC3988254
- DOI: 10.1016/j.jss.2014.01.011
Membrane potential-dependent uptake of 18F-triphenylphosphonium--a new voltage sensor as an imaging agent for detecting burn-induced apoptosis
Abstract
Background: Mitochondrial dysfunction has been closely related to many pathologic processes, such as cellular apoptosis. Alterations in organelle membrane potential are associated with mitochondrial dysfunction. A fluorine-18 labeled phosphonium compound: (18)F-triphenylphosphonium ((18)F-TPP) was prepared to determine its potential use as a mitochondria-targeting radiopharmaceutical to evaluate cellular apoptosis.
Methods: Studies were conducted in both ex vivo cell lines and in vivo using a burned animal model. Uptake of (18)F-TPP was assessed in PC-3 cells by gamma counting under the following conditions: graded levels of extracellular potassium concentrations, incubation with carbonyl cyanide m-chlorophenylhydrazone and staurosporine. Apoptosis was studied in a burn animal model using terminal deoxynucleotidyl transferase dUTP nick end labeling staining and simultaneous assessment of (18)F-TPP uptake by biodistribution.
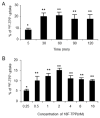
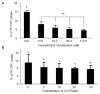
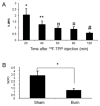
Results: We found that stepwise membrane depolarization by potassium (K) resulted in a linear decrease in (18)F-TPP uptake, with a slope of 0.62 ± 0.08 and a correlation coefficient of 0.936 ± 0.11. Gradually increased concentrations of m-chlorophenylhydrazone lead to decreased uptake of (18)F-TPP. Staurosporine significantly decreased the uptake of (18)F-TPP in PC-3 cells from 14.2 ± 3.8% to 5.6 ± 1.3% (P < 0.001). Burn-induced significant apoptosis (sham: 4.4 ± 1.8% versus burn: 24.6 ± 6.7 %; P < 0.005) and a reduced uptake of tracer in the spleens of burn-injured animals as compared with sham burn controls (burn: 1.13 ± 0.24% versus sham: 3.28 ± 0.67%; P < 0.005). Biodistribution studies demonstrated that burn-induced significant reduction in (18)F-TPP uptake in spleen, heart, lung, and liver, which were associated with significantly increased apoptosis.
Conclusions: (18)F-TPP is a promising new voltage sensor for detecting mitochondrial dysfunction and apoptosis in various tissues.
Keywords: (18)F-TPP; Apoptosis; Membrane potential; Mitochondria.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures


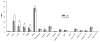
Similar articles
-
Characterization of membrane potential-dependent uptake of the novel PET tracer 18F-fluorobenzyl triphenylphosphonium cation.Eur J Nucl Med Mol Imaging. 2007 Dec;34(12):2057-65. doi: 10.1007/s00259-007-0500-8. Epub 2007 Sep 5. Eur J Nucl Med Mol Imaging. 2007. PMID: 17786439
-
4-[18F]-tetraphenylphosphonium as a PET tracer for myocardial mitochondrial membrane potential.JACC Cardiovasc Imaging. 2012 Mar;5(3):285-92. doi: 10.1016/j.jcmg.2011.11.017. JACC Cardiovasc Imaging. 2012. PMID: 22421174
-
Synthesis of [18F]-labeled (2-(2-fluoroethoxy)ethyl)tris(4-methoxyphenyl)phosphonium cation as a potential agent for positron emission tomography myocardial imaging.Nucl Med Biol. 2012 Oct;39(7):1093-8. doi: 10.1016/j.nucmedbio.2012.03.008. Epub 2012 May 8. Nucl Med Biol. 2012. PMID: 22575270
-
Delocalized Lipophilic Cation Triphenyl Phosphonium: Promising Molecule for Mitochondria Targeting.Curr Drug Deliv. 2023;20(9):1217-1223. doi: 10.2174/1567201819666220525092527. Curr Drug Deliv. 2023. PMID: 35619273 Review.
-
Quantification of Myocardial Mitochondrial Membrane Potential Using PET.Curr Cardiol Rep. 2021 May 10;23(6):70. doi: 10.1007/s11886-021-01500-8. Curr Cardiol Rep. 2021. PMID: 33970353 Free PMC article. Review.
Cited by
-
Mitochondria-Targeted Triphenylphosphonium-Based Compounds: Syntheses, Mechanisms of Action, and Therapeutic and Diagnostic Applications.Chem Rev. 2017 Aug 9;117(15):10043-10120. doi: 10.1021/acs.chemrev.7b00042. Epub 2017 Jun 27. Chem Rev. 2017. PMID: 28654243 Free PMC article. Review.
-
Biomarkers in preclinical cancer imaging.Eur J Nucl Med Mol Imaging. 2015 Apr;42(4):579-96. doi: 10.1007/s00259-014-2980-7. Epub 2015 Feb 12. Eur J Nucl Med Mol Imaging. 2015. PMID: 25673052 Free PMC article. Review.
-
Mitochondrial-Targeted Molecular Imaging in Cardiac Disease.Biomed Res Int. 2017;2017:5246853. doi: 10.1155/2017/5246853. Epub 2017 May 30. Biomed Res Int. 2017. PMID: 28638829 Free PMC article. Review.
References
-
- Zheng M, Zhang Q, Joe Y, et al. Curcumin induces apoptotic cell death of activated human CD4+ T cells via increasing endoplasmic reticulum stress and mitochondrial dysfunction. Int Immunopharmacol. 2013;15:517. - PubMed
-
- Hwang KE, Park C, Kwon SJ, et al. Synergistic induction of apoptosis by sulindac and simvastatin in A549 human lung cancer cells via reactive oxygen species-dependent mitochondrial dysfunction. Int J Oncol. 2013;43:262. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

