Reactive oxygen species contribute to arsenic-induced EZH2 phosphorylation in human bronchial epithelial cells and lung cancer cells
- PMID: 24582688
- PMCID: PMC4401465
- DOI: 10.1016/j.taap.2014.02.005
Reactive oxygen species contribute to arsenic-induced EZH2 phosphorylation in human bronchial epithelial cells and lung cancer cells
Abstract
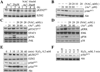
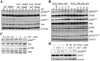
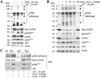
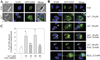
Our previous studies suggested that arsenic is able to induce serine 21 phosphorylation of the EZH2 protein through activation of JNK, STAT3, and Akt signaling pathways in the bronchial epithelial cell line, BEAS-2B. In the present report, we further demonstrated that reactive oxygen species (ROS) were involved in the arsenic-induced protein kinase activation that leads to EZH2 phosphorylation. Several lines of evidence supported this notion. First, the pretreatment of the cells with N-acetyl-l-cysteine (NAC), a potent antioxidant, abolishes arsenic-induced EZH2 phosphorylation along with the inhibition of JNK, STAT3, and Akt. Second, H2O2, the most important form of ROS in the cells in response to extracellular stress signals, can induce phosphorylation of the EZH2 protein and the activation of JNK, STAT3, and Akt. By ectopic expression of the myc-tagged EZH2, we additionally identified direct interaction and phosphorylation of the EZH2 protein by Akt in response to arsenic and H2O2. Furthermore, both arsenic and H2O2 were able to induce the translocation of ectopically expressed or endogenous EZH2 from nucleus to cytoplasm. In summary, the data presented in this report indicate that oxidative stress due to ROS generation plays an important role in the arsenic-induced EZH2 phosphorylation.
Keywords: Akt; Arsenic; Cytoplasmic translocation; EZH2; Oxidative stress; Phosphorylation.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
JNK and STAT3 signaling pathways converge on Akt-mediated phosphorylation of EZH2 in bronchial epithelial cells induced by arsenic.Cell Cycle. 2013 Jan 1;12(1):112-21. doi: 10.4161/cc.23030. Epub 2012 Dec 19. Cell Cycle. 2013. PMID: 23255093 Free PMC article.
-
EZH2 phosphorylation regulates Tat-induced HIV-1 transactivation via ROS/Akt signaling pathway.FEBS Lett. 2015 Dec 21;589(24 Pt B):4106-11. doi: 10.1016/j.febslet.2015.11.033. Epub 2015 Nov 26. FEBS Lett. 2015. PMID: 26632198
-
Linking JNK-STAT3-Akt signaling axis to EZH2 phosphorylation: a novel pathway of carcinogenesis.Cell Cycle. 2013 Jan 15;12(2):202-3. doi: 10.4161/cc.23419. Epub 2012 Jan 15. Cell Cycle. 2013. PMID: 23287465 Free PMC article. No abstract available.
-
Oncogenic transformation of human lung bronchial epithelial cells induced by arsenic involves ROS-dependent activation of STAT3-miR-21-PDCD4 mechanism.Sci Rep. 2016 Nov 23;6:37227. doi: 10.1038/srep37227. Sci Rep. 2016. Retraction in: Sci Rep. 2019 Nov 15;9(1):17001. doi: 10.1038/s41598-019-53858-z. PMID: 27876813 Free PMC article. Retracted.
-
Advances in bi-directional relationships for EZH2 and oxidative stress.Exp Cell Res. 2024 Jan 1;434(1):113876. doi: 10.1016/j.yexcr.2023.113876. Epub 2023 Dec 7. Exp Cell Res. 2024. PMID: 38070859 Review.
Cited by
-
Epigallocatechin-3-gallate enhances the therapeutic effects of leptomycin B on human lung cancer a549 cells.Oxid Med Cell Longev. 2015;2015:217304. doi: 10.1155/2015/217304. Epub 2015 Apr 1. Oxid Med Cell Longev. 2015. PMID: 25922640 Free PMC article.
-
Activation of SAPK/JNK mediated the inhibition and reciprocal interaction of DNA methyltransferase 1 and EZH2 by ursolic acid in human lung cancer cells.J Exp Clin Cancer Res. 2015 Sep 11;34(1):99. doi: 10.1186/s13046-015-0215-9. J Exp Clin Cancer Res. 2015. PMID: 26362062 Free PMC article.
-
Carnosic Acid, a Natural Diterpene, Attenuates Arsenic-Induced Hepatotoxicity via Reducing Oxidative Stress, MAPK Activation, and Apoptotic Cell Death Pathway.Oxid Med Cell Longev. 2018 May 2;2018:1421438. doi: 10.1155/2018/1421438. eCollection 2018. Oxid Med Cell Longev. 2018. PMID: 29854073 Free PMC article.
-
Arsenic induces polyadenylation of canonical histone mRNA by down-regulating stem-loop-binding protein gene expression.J Biol Chem. 2014 Nov 14;289(46):31751-31764. doi: 10.1074/jbc.M114.591883. Epub 2014 Sep 28. J Biol Chem. 2014. PMID: 25266719 Free PMC article.
-
Oxidative stress, epigenetics, and cancer stem cells in arsenic carcinogenesis and prevention.Curr Pharmacol Rep. 2016 Apr;2(2):57-63. doi: 10.1007/s40495-016-0049-y. Epub 2016 Jan 23. Curr Pharmacol Rep. 2016. PMID: 27134817 Free PMC article.
References
-
- Ahmad S, Kitchin KT, Cullen WR. Arsenic species that cause release of iron from ferritin and generation of activated oxygen. Arch. Biochem. Biophys. 2000;382:195–202. - PubMed
-
- Alessi DR, Caudwell FB, Andjelkovic M, Hemmings BA, Cohen P. Molecular basis for the substrate specificity of protein kinase B; comparison with MAPKAP kinase-1 and p70 S6 kinase. FEBS Lett. 1996;399:333–338. - PubMed
-
- Cha TL, Zhou BP, Xia W, Wu Y, Yang CC, Chen CT, Ping B, Otte AP, Hung MC. Akt-mediated phosphorylation of EZH2 suppresses methylation of lysine 27 in histone H3. Science. 2005;310:306–310. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

