μ-Opioid receptor inhibition of substance P release from primary afferents disappears in neuropathic pain but not inflammatory pain
- PMID: 24583035
- PMCID: PMC3998911
- DOI: 10.1016/j.neuroscience.2014.02.023
μ-Opioid receptor inhibition of substance P release from primary afferents disappears in neuropathic pain but not inflammatory pain
Abstract
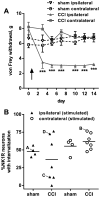
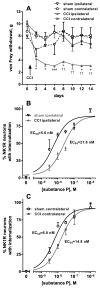
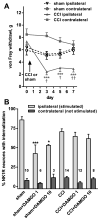
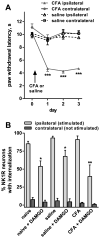
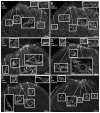
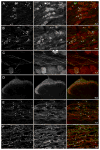
Opiate analgesia in the spinal cord is impaired during neuropathic pain. We hypothesized that this is caused by a decrease in μ-opioid receptor inhibition of neurotransmitter release from primary afferents. To investigate this possibility, we measured substance P release in the spinal dorsal horn as neurokinin 1 receptor (NK1R) internalization in rats with chronic constriction injury (CCI) of the sciatic nerve. Noxious stimulation of the paw with CCI produced inconsistent NK1R internalization, suggesting that transmission of nociceptive signals by the injured nerve was variably impaired after CCI. This idea was supported by the fact that CCI produced only small changes in the ability of exogenous substance P to induce NK1R internalization or in the release of substance P evoked centrally from site of nerve injury. In subsequent experiments, NK1R internalization was induced in spinal cord slices by stimulating the dorsal root ipsilateral to CCI. We observed a complete loss of the inhibition of substance P release by the μ-opioid receptor agonist [D-Ala(2), NMe-Phe(4), Gly-ol(5)]-enkephalin (DAMGO) in CCI rats but not in sham-operated rats. In contrast, DAMGO still inhibited substance P release after inflammation of the hind paw with complete Freund's adjuvant and in naïve rats. This loss of inhibition was not due to μ-opioid receptor downregulation in primary afferents, because their colocalization with substance P was unchanged, both in dorsal root ganglion neurons and primary afferent fibers in the dorsal horn. In conclusion, nerve injury eliminates the inhibition of substance P release by μ-opioid receptors, probably by hindering their signaling mechanisms.
Keywords: inflammation; internalization; neurokinin 1 receptor; neuropathic pain; opioid receptor; substance P.
Published by Elsevier Ltd.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

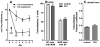
Similar articles
-
Inhibition by spinal mu- and delta-opioid agonists of afferent-evoked substance P release.J Neurosci. 2005 Apr 6;25(14):3651-60. doi: 10.1523/JNEUROSCI.0252-05.2005. J Neurosci. 2005. PMID: 15814796 Free PMC article.
-
Inflammation enhances Y1 receptor signaling, neuropeptide Y-mediated inhibition of hyperalgesia, and substance P release from primary afferent neurons.Neuroscience. 2014 Jan 3;256:178-94. doi: 10.1016/j.neuroscience.2013.10.054. Epub 2013 Oct 31. Neuroscience. 2014. PMID: 24184981 Free PMC article.
-
Src family kinases mediate the inhibition of substance P release in the rat spinal cord by μ-opioid receptors and GABA(B) receptors, but not α2 adrenergic receptors.Eur J Neurosci. 2010 Sep;32(6):963-73. doi: 10.1111/j.1460-9568.2010.07335.x. Epub 2010 Aug 19. Eur J Neurosci. 2010. PMID: 20726886 Free PMC article.
-
Opioid receptor trafficking and interaction in nociceptors.Br J Pharmacol. 2015 Jan;172(2):364-74. doi: 10.1111/bph.12653. Epub 2014 Jul 1. Br J Pharmacol. 2015. PMID: 24611685 Free PMC article. Review.
-
Drug-specific differences in the ability of opioids to manage burn pain.Burns. 2020 May;46(3):503-513. doi: 10.1016/j.burns.2019.03.028. Epub 2019 Dec 16. Burns. 2020. PMID: 31859093 Review.
Cited by
-
Impact of physical activity on pain perception in an animal model of endometriosis.J Endometr Pelvic Pain Disord. 2015 Jul-Sep;7(3):89-114. doi: 10.5301/je.5000231. Epub 2015 Dec 24. J Endometr Pelvic Pain Disord. 2015. PMID: 28217664 Free PMC article.
-
Neuropeptide Y release in the rat spinal cord measured with Y1 receptor internalization is increased after nerve injury.Neuropharmacology. 2019 Nov 1;158:107732. doi: 10.1016/j.neuropharm.2019.107732. Epub 2019 Aug 2. Neuropharmacology. 2019. PMID: 31377198 Free PMC article.
-
Neurokinin 1 and opioid receptors: relationships and interactions in nervous system.Transl Perioper Pain Med. 2016;1(3):11-21. Transl Perioper Pain Med. 2016. PMID: 28409174 Free PMC article.
-
Transmission pathways and mediators as the basis for clinical pharmacology of pain.Expert Rev Clin Pharmacol. 2016 Oct;9(10):1363-1387. doi: 10.1080/17512433.2016.1204231. Epub 2016 Jul 4. Expert Rev Clin Pharmacol. 2016. PMID: 27322358 Free PMC article.
-
Plasma Concentration of Norepinephrine, β-endorphin, and Substance P in Lame Dairy Cows.J Vet Res. 2018 Oct 24;62(2):193-197. doi: 10.2478/jvetres-2018-0029. eCollection 2018 Jun. J Vet Res. 2018. PMID: 30364907 Free PMC article.
References
-
- Aimone LD, Jones SL, Gebhart GF. Stimulation-produced descending inhibition from the periaqueductal gray and nucleus raphe magnus in the rat: mediation by spinal monoamines but not opioids. Pain. 1987;31:123–136. - PubMed
-
- Allen BJ, Li J, Menning PM, Rogers SD, Ghilardi J, Mantyh PW, Simone DA. Primary afferent fibers that contribute to increased substance P receptor internalization in the spinal cord after injury. J Neurophysiol. 1999;81:1379–1390. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

